Systematic Overview: Efficacy of Pharmacotherapy and Psychotherapy
VerifiedAdded on 2023/06/11
|10
|11051
|428
Literature Review
AI Summary
This review systematically examines the efficacy of pharmacotherapy and psychotherapy for major psychiatric disorders in adults. It analyzes 61 meta-analyses encompassing 852 individual trials and 137,126 participants, comparing the effectiveness of these treatments against placebo, head-to-head, and in combination. The review also assesses the methodological quality of the included trials. Findings indicate that both pharmacotherapies and psychotherapies are effective, with psychotherapies showing higher effect sizes compared to placebo, though direct comparisons lack consistent differences. Pharmacotherapy trials generally exhibit larger sample sizes and better blinding, while psychotherapy trials report lower dropout rates and follow-up data. The review highlights the need for well-designed direct comparisons and research into combining both treatment modalities for optimal patient outcomes. It also addresses the methodological differences between pharmacotherapy and psychotherapy trials, emphasizing the limitations of indirect comparisons.

Copyright 2014 American Medical Association. All rights reserved.
Efficacy of Pharmacotherapy and Psychotherapy
for Adult Psychiatric Disorders
A Systematic Overview of Meta-analyses
Maximilian Huhn, MD; Magdolna Tardy, MSc; Loukia Maria Spineli, MSc; Werner Kissling, MD; Hans Förstl, MD;
Gabriele Pitschel-Walz, PhD; Claudia Leucht, MD; Myrto Samara, MD; Markus Dold, MD; John M. Davis, MD;
Stefan Leucht, MD
IMPORTANCEThere is debate about the effectiveness of psychiatric treatments and whether
pharmacotherapy or psychotherapy should be primarily used.
OBJECTIVESTo perform a systematic overview on the efficacy of pharmacotherapies and
psychotherapies for major psychiatric disorders and to compare the quality of
pharmacotherapy and psychotherapy trials.
EVIDENCE REVIEWWe searched MEDLINE, EMBASE, PsycINFO, and the Cochrane Library
(April 2012, with no time or language limit) for systematic reviews on pharmacotherapy or
psychotherapy vs placebo, pharmacotherapy vs psychotherapy, and their combination vs
either modality alone. Two reviewers independently selected the meta-analyses and
extracted efficacy effect sizes. We assessed the quality of the individual trials included in the
pharmacotherapy and psychotherapy meta-analyses with the Cochrane risk of bias tool.
FINDINGSThe search yielded 45 233 results. We included 61 meta-analyses on 21 psychiatric
disorders, which contained 852 individual trials and 137 126 participants. The mean effect size
of the meta-analyses was medium (mean, 0.50; 95% CI, 0.41-0.59). Effect sizes of
psychotherapies vs placebo tended to be higher than those of medication, but direct
comparisons, albeit usually based on few trials, did not reveal consistent differences.
Individual pharmacotherapy trials were more likely to have large sample sizes, blinding,
control groups, and intention-to-treat analyses. In contrast, psychotherapy trials had lower
dropout rates and provided follow-up data. In psychotherapy studies, wait-list designs
showed larger effects than did comparisons with placebo.
CONCLUSIONS AND RELEVANCEMany pharmacotherapies and psychotherapies are effective,
but there is a lot of room for improvement. Because of the multiple differences in the
methods used in pharmacotherapy and psychotherapy trials, indirect comparisons of their
effect sizes compared with placebo or no treatment are problematic. Well-designed direct
comparisons, which are scarce, need public funding. Because patients often benefit from
both forms of therapy, research should also focus on how both modalities can be best
combined to maximize synergy rather than debate the use of one treatment over the other.
JAMA Psychiatry. 2014;71(6):706-715. doi:10.1001/jamapsychiatry.2014.112
Published online April 30, 2014.
Editorial page 624
Supplemental content at
jamapsychiatry.com
Author Affiliations: Department of
Psychiatry and Psychotherapy,
Technische Universität München,
München, Germany (Huhn, Tardy,
Kissling, Förstl, Pitschel-Walz, Claudia
Leucht, Samara, Dold, S. Leucht);
Department of Psychiatry, University
of Oxford, Warneford Hospital,
Oxford, England (C. Leucht, Stefan
Leucht); Department of Hygiene and
Epidemiology, School of Medicine,
University of Ioannina, University
Campus, Ioannina, Greece (Spineli);
Psychiatric Institute, University of
Illinois at Chicago (Davis); Institute of
Psychiatry, King’s College London,
London, England (S. Leucht).
Corresponding Author: Stefan
Leucht, MD, Department of
Psychiatry and Psychotherapy,
Technische Universität München,
Klinikum rechts der Isar,
Ismaningerstrasse 22, 81675
München, Germany (stefan.leucht
@lrz.tum.de).
Clinical Review & Education
Review
706 jamapsychiatry.com
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
Efficacy of Pharmacotherapy and Psychotherapy
for Adult Psychiatric Disorders
A Systematic Overview of Meta-analyses
Maximilian Huhn, MD; Magdolna Tardy, MSc; Loukia Maria Spineli, MSc; Werner Kissling, MD; Hans Förstl, MD;
Gabriele Pitschel-Walz, PhD; Claudia Leucht, MD; Myrto Samara, MD; Markus Dold, MD; John M. Davis, MD;
Stefan Leucht, MD
IMPORTANCEThere is debate about the effectiveness of psychiatric treatments and whether
pharmacotherapy or psychotherapy should be primarily used.
OBJECTIVESTo perform a systematic overview on the efficacy of pharmacotherapies and
psychotherapies for major psychiatric disorders and to compare the quality of
pharmacotherapy and psychotherapy trials.
EVIDENCE REVIEWWe searched MEDLINE, EMBASE, PsycINFO, and the Cochrane Library
(April 2012, with no time or language limit) for systematic reviews on pharmacotherapy or
psychotherapy vs placebo, pharmacotherapy vs psychotherapy, and their combination vs
either modality alone. Two reviewers independently selected the meta-analyses and
extracted efficacy effect sizes. We assessed the quality of the individual trials included in the
pharmacotherapy and psychotherapy meta-analyses with the Cochrane risk of bias tool.
FINDINGSThe search yielded 45 233 results. We included 61 meta-analyses on 21 psychiatric
disorders, which contained 852 individual trials and 137 126 participants. The mean effect size
of the meta-analyses was medium (mean, 0.50; 95% CI, 0.41-0.59). Effect sizes of
psychotherapies vs placebo tended to be higher than those of medication, but direct
comparisons, albeit usually based on few trials, did not reveal consistent differences.
Individual pharmacotherapy trials were more likely to have large sample sizes, blinding,
control groups, and intention-to-treat analyses. In contrast, psychotherapy trials had lower
dropout rates and provided follow-up data. In psychotherapy studies, wait-list designs
showed larger effects than did comparisons with placebo.
CONCLUSIONS AND RELEVANCEMany pharmacotherapies and psychotherapies are effective,
but there is a lot of room for improvement. Because of the multiple differences in the
methods used in pharmacotherapy and psychotherapy trials, indirect comparisons of their
effect sizes compared with placebo or no treatment are problematic. Well-designed direct
comparisons, which are scarce, need public funding. Because patients often benefit from
both forms of therapy, research should also focus on how both modalities can be best
combined to maximize synergy rather than debate the use of one treatment over the other.
JAMA Psychiatry. 2014;71(6):706-715. doi:10.1001/jamapsychiatry.2014.112
Published online April 30, 2014.
Editorial page 624
Supplemental content at
jamapsychiatry.com
Author Affiliations: Department of
Psychiatry and Psychotherapy,
Technische Universität München,
München, Germany (Huhn, Tardy,
Kissling, Förstl, Pitschel-Walz, Claudia
Leucht, Samara, Dold, S. Leucht);
Department of Psychiatry, University
of Oxford, Warneford Hospital,
Oxford, England (C. Leucht, Stefan
Leucht); Department of Hygiene and
Epidemiology, School of Medicine,
University of Ioannina, University
Campus, Ioannina, Greece (Spineli);
Psychiatric Institute, University of
Illinois at Chicago (Davis); Institute of
Psychiatry, King’s College London,
London, England (S. Leucht).
Corresponding Author: Stefan
Leucht, MD, Department of
Psychiatry and Psychotherapy,
Technische Universität München,
Klinikum rechts der Isar,
Ismaningerstrasse 22, 81675
München, Germany (stefan.leucht
@lrz.tum.de).
Clinical Review & Education
Review
706 jamapsychiatry.com
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

Copyright 2014 American Medical Association. All rights reserved.
M uch controversy surrounds psychiatry, including a de-
bate about the effectiveness of its treatments. For ex-
ample, the efficacy of antipsychotics,
1
cholinesterase
inhibitors,2and lithium prophylaxis3has been questioned, and Kirsch
et al4 concluded that antidepressants should be used only in se-
verely ill patients. Leucht et al5 showed that psychiatric drugs have
the same range of efficacy as frequently used drugs from other medi-
cal specialties. Nevertheless, the criticism has expanded to psycho-
therapy, in particular psychoanalysis,6 but also to cognitive behav-
ioral therapy (CBT).7 Moreover,whether psychotherapy or
medication should be used to treat psychiatric conditions is strongly
disputed. This ongoing criticism of psychiatry and the debate about
the appropriate treatment make it necessary to examine the situa-
tion and present a broad appraisal. Such an attempt has been made
possible by meta-analyses, which provide standardized quantita-
tive measures of their efficacy by the calculation of effect sizes.8 We
therefore conducted a systematic overview of meta-analyses on the
efficacy of drug therapy and psychotherapy for major psychiatric dis-
orders, which we classified in 3 comparisons: psychotherapy or phar-
macotherapy compared with placebo or no treatment, head-to-
head comparisons of pharmacotherapies and psychotherapies, and
the combination of both treatments. Moreover, we compared the
methodologic quality of the individual drug and psychotherapy trials
included in these meta-analyses. We had 3 objectives. First, we aimed
to present an overview of what benefits psychiatry as a field can pro-
vide for patients, examining the contributions of pharmaco-
therapy and psychotherapy. Second, we wanted to determine the
understudied areas, which can be roughly derived from the num-
bers of studies and participants included in the meta-analyses. Fi-
nally, we examined whether pharmacotherapy and psychotherapy
trials have general methodologic differences that would be impor-
tant for the interpretation of their meta-analyses. We emphasize that,
because of obvious limitations, our overview is not a guideline of
which treatments should be used and which should not be used for
a disorder. Nevertheless, the systematic approach should lead to re-
sults with important implications for psychiatry.
Methods
Identification of Diseases and Search Strategy
We drafted a protocol and made it freely available on our institu-
tional website (http://tinyurl.com/d6b277p; Supplement [eAppen-
dix 1]).9 Two authors (M.H. and S.L.) selected major psychiatric dis-
orders by reviewing the DSM-IV and InternationalStatistical
Classification of Diseases, 10th Revision (ICD-10) (Supplement [eAp-
pendix 1]). We did not examine child psychiatric disorders (ICD-10
categories F80-F98 and DSM-IV categories 307-319).
We searched PubMed, EMBASE, and PsycINFO (last search on
April 22, 2012; no time or language limit) and individual records of
the Cochrane Library for systematic reviews and meta-analyses of
randomized clinical trials (RCTs) on the following comparisons: (1)
pharmacotherapy or psychotherapy vs placebo, (2) head-to-head
pharmacotherapy vs psychotherapy, and (3) combinations of phar-
macotherapy and psychotherapy vs monotherapy with each inter-
vention. Search terms were meta-analy* or metaanaly* or system-
atic review* combined with the thesaurus of the individual databases
concerning each disorder (Supplement [eAppendix 2]). Titles, ab-
stracts, and, subsequently, full articles were examined by one au-
thor (M.H.), and another (M.T.) independently examined a random
sample of 20% of the retrieved publications. Disagreements were
resolved by discussion with a third author (S.L.). Only between-
group effect sizes were extracted; within-treatment effect sizes were
not used because they generally inflate effects. We included only
standard treatments (eg, antipsychotics for schizophrenia [not
lithium]) and aimed to include one meta-analysis for each disorder.
We applied the following a priori–defined criteria to operationalize
the selection: (1) all patients rather than special populations (eg, only
first-episode patients); (2) classes of drugs rather than single drugs
(eg, any antipsychotic rather than only haloperidol); (3) CBT and psy-
chodynamic approaches (the former are usually recommended by
guidelines; we checked the National Institute for Health and Clini-
cal Excellence10and noted that psychodynamic therapy is consid-
ered a counterpart to CBT) unless other treatments are the stan-
dard (eg, cognitive training for dementia); when reviews on these
specific psychotherapies were not available, we used meta-
analyses on all psychotherapies; (4) the most up-to-date findings;
and (5) full reporting of the results. In case of doubt, Cochrane re-
views were preferred.
Parameters Extracted From the Meta-analyses
Two authors (M.H. and M.T.) independently extracted results on the
primary efficacy outcomes, which generally were the mean (change)
scores of overall symptoms at the end point (eg, Hamilton Scale for D
pression) and study-defined responder rates (for acute treatment) or
relapse rates (for maintenance treatment). We also recorded the num
ber of studies and participants included in the meta-analyses and the
mean study duration and number of psychotherapy sessions.
Dichotomous Outcomes
We extracted the percentage of responders (or relapsers) in drug and
placebo groups, the response ratio or relative risk reduction (both ab-
breviated as RR), the absolute response or risk difference (ARD), and
the number needed to treat (NNT), all with their 95% CIs. The Supple
ment (eAppendix 3) presents a detailed explanation.
Continuous Outcomes
For mean values of rating scales, we recorded between-group mean
differences (MDs) (ie, the difference between the raw scores) and
standardized MDs (SMDs) (effect size, Hedges g8 value if avail-
able), which express MD in standard deviation (SD) units. We fol-
lowed the original authors’ decisions concerning fixed- vs random-
effects models (the general use of a random-effects model did not
change the overall results [Supplement; eAppendix 4 and eTable 4]).
Missing Parameters
If necessary, we transformed the data to our 5 parameters (MD/SMD/
ARD/RR/NNT) or recalculated meta-analyses by entering single-trial
results into meta-analytic software programs.11,12
When only effect sizes
for dichotomous outcomes (ARD, RR) were available, SMDs were es-
timated with Comprehensive Meta-analysis, version 2.12The pur-
pose was to present all results in a single unit (SMD) in the figures.
Extraction of Quality Indicators From Individual Studies
One author (M.H., C.L., M.S., or M.D.) retrieved all studies included
in the meta-analyses (only the acute phase to enhance comparabil-
Pharmacotherapy and Psychotherapy in Adults Review Clinical Review & Education
jamapsychiatry.com JAMA PsychiatryJune 2014Volume 71, Number 6707
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
M uch controversy surrounds psychiatry, including a de-
bate about the effectiveness of its treatments. For ex-
ample, the efficacy of antipsychotics,
1
cholinesterase
inhibitors,2and lithium prophylaxis3has been questioned, and Kirsch
et al4 concluded that antidepressants should be used only in se-
verely ill patients. Leucht et al5 showed that psychiatric drugs have
the same range of efficacy as frequently used drugs from other medi-
cal specialties. Nevertheless, the criticism has expanded to psycho-
therapy, in particular psychoanalysis,6 but also to cognitive behav-
ioral therapy (CBT).7 Moreover,whether psychotherapy or
medication should be used to treat psychiatric conditions is strongly
disputed. This ongoing criticism of psychiatry and the debate about
the appropriate treatment make it necessary to examine the situa-
tion and present a broad appraisal. Such an attempt has been made
possible by meta-analyses, which provide standardized quantita-
tive measures of their efficacy by the calculation of effect sizes.8 We
therefore conducted a systematic overview of meta-analyses on the
efficacy of drug therapy and psychotherapy for major psychiatric dis-
orders, which we classified in 3 comparisons: psychotherapy or phar-
macotherapy compared with placebo or no treatment, head-to-
head comparisons of pharmacotherapies and psychotherapies, and
the combination of both treatments. Moreover, we compared the
methodologic quality of the individual drug and psychotherapy trials
included in these meta-analyses. We had 3 objectives. First, we aimed
to present an overview of what benefits psychiatry as a field can pro-
vide for patients, examining the contributions of pharmaco-
therapy and psychotherapy. Second, we wanted to determine the
understudied areas, which can be roughly derived from the num-
bers of studies and participants included in the meta-analyses. Fi-
nally, we examined whether pharmacotherapy and psychotherapy
trials have general methodologic differences that would be impor-
tant for the interpretation of their meta-analyses. We emphasize that,
because of obvious limitations, our overview is not a guideline of
which treatments should be used and which should not be used for
a disorder. Nevertheless, the systematic approach should lead to re-
sults with important implications for psychiatry.
Methods
Identification of Diseases and Search Strategy
We drafted a protocol and made it freely available on our institu-
tional website (http://tinyurl.com/d6b277p; Supplement [eAppen-
dix 1]).9 Two authors (M.H. and S.L.) selected major psychiatric dis-
orders by reviewing the DSM-IV and InternationalStatistical
Classification of Diseases, 10th Revision (ICD-10) (Supplement [eAp-
pendix 1]). We did not examine child psychiatric disorders (ICD-10
categories F80-F98 and DSM-IV categories 307-319).
We searched PubMed, EMBASE, and PsycINFO (last search on
April 22, 2012; no time or language limit) and individual records of
the Cochrane Library for systematic reviews and meta-analyses of
randomized clinical trials (RCTs) on the following comparisons: (1)
pharmacotherapy or psychotherapy vs placebo, (2) head-to-head
pharmacotherapy vs psychotherapy, and (3) combinations of phar-
macotherapy and psychotherapy vs monotherapy with each inter-
vention. Search terms were meta-analy* or metaanaly* or system-
atic review* combined with the thesaurus of the individual databases
concerning each disorder (Supplement [eAppendix 2]). Titles, ab-
stracts, and, subsequently, full articles were examined by one au-
thor (M.H.), and another (M.T.) independently examined a random
sample of 20% of the retrieved publications. Disagreements were
resolved by discussion with a third author (S.L.). Only between-
group effect sizes were extracted; within-treatment effect sizes were
not used because they generally inflate effects. We included only
standard treatments (eg, antipsychotics for schizophrenia [not
lithium]) and aimed to include one meta-analysis for each disorder.
We applied the following a priori–defined criteria to operationalize
the selection: (1) all patients rather than special populations (eg, only
first-episode patients); (2) classes of drugs rather than single drugs
(eg, any antipsychotic rather than only haloperidol); (3) CBT and psy-
chodynamic approaches (the former are usually recommended by
guidelines; we checked the National Institute for Health and Clini-
cal Excellence10and noted that psychodynamic therapy is consid-
ered a counterpart to CBT) unless other treatments are the stan-
dard (eg, cognitive training for dementia); when reviews on these
specific psychotherapies were not available, we used meta-
analyses on all psychotherapies; (4) the most up-to-date findings;
and (5) full reporting of the results. In case of doubt, Cochrane re-
views were preferred.
Parameters Extracted From the Meta-analyses
Two authors (M.H. and M.T.) independently extracted results on the
primary efficacy outcomes, which generally were the mean (change)
scores of overall symptoms at the end point (eg, Hamilton Scale for D
pression) and study-defined responder rates (for acute treatment) or
relapse rates (for maintenance treatment). We also recorded the num
ber of studies and participants included in the meta-analyses and the
mean study duration and number of psychotherapy sessions.
Dichotomous Outcomes
We extracted the percentage of responders (or relapsers) in drug and
placebo groups, the response ratio or relative risk reduction (both ab-
breviated as RR), the absolute response or risk difference (ARD), and
the number needed to treat (NNT), all with their 95% CIs. The Supple
ment (eAppendix 3) presents a detailed explanation.
Continuous Outcomes
For mean values of rating scales, we recorded between-group mean
differences (MDs) (ie, the difference between the raw scores) and
standardized MDs (SMDs) (effect size, Hedges g8 value if avail-
able), which express MD in standard deviation (SD) units. We fol-
lowed the original authors’ decisions concerning fixed- vs random-
effects models (the general use of a random-effects model did not
change the overall results [Supplement; eAppendix 4 and eTable 4]).
Missing Parameters
If necessary, we transformed the data to our 5 parameters (MD/SMD/
ARD/RR/NNT) or recalculated meta-analyses by entering single-trial
results into meta-analytic software programs.11,12
When only effect sizes
for dichotomous outcomes (ARD, RR) were available, SMDs were es-
timated with Comprehensive Meta-analysis, version 2.12The pur-
pose was to present all results in a single unit (SMD) in the figures.
Extraction of Quality Indicators From Individual Studies
One author (M.H., C.L., M.S., or M.D.) retrieved all studies included
in the meta-analyses (only the acute phase to enhance comparabil-
Pharmacotherapy and Psychotherapy in Adults Review Clinical Review & Education
jamapsychiatry.com JAMA PsychiatryJune 2014Volume 71, Number 6707
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015

Copyright 2014 American Medical Association. All rights reserved.
ity) and assessed their quality with the Cochrane risk of bias tool,13
which is based on scientific evidence of associations between over-
estimates of effect and methodologic shortcomings in the follow-
ing domains:
1. appropriate randomization (eg, computer-generated random-
ization sequence),
2. allocation concealment (eg, voice mail system),
3. blinding (because blinding of therapists is impossible in psycho-
therapy trials, we compared blind outcome assessment with no
blinding), and
4. missing outcomes (whether patients who withdrew from the
study and their reasons for withdrawal were reported, an inten-
tion-to-treat analysis was performed, and we also reported the
size of the overall dropout rate).
We did not examine the tool’s domain selective reporting but
added an item to the control groups that we classified as any con-
temporaneous treatment (placebo, treatment as usual, or ineffec-
tive therapy) vs wait list or no treatment. All items were rated as low,
unclear, and high risk of bias, with items rated as unclear (eg, indi-
cated as randomized without further explanation) combined with
those having a high risk of bias in the statistical analysis. Moreover,
we analyzed how often follow-up data after the trial end point were
collected.
Statistical Analysis
Statistical analysis was conducted in 4 steps.
1. We compared the sample sizes of psychotherapy and pharma-
cotherapy meta-analyses and their quality as measured by A Mea-
surement Tool to Assess Systematic Reviews (AMSTAR) score.14
2. We compared the sample sizes and the methodologic quality of
the individualacute-phase studies included in the psycho-
therapy and pharmacotherapy meta-analyses with the Coch-
rane risk of bias tool.13The same analysis was performed on the
single disorders (Supplement [eAppendix 4, eFigures 1-7]).
3. We examined whether the meta-analyses conducted subgroup
comparisons on the risk of bias tool domains (eg, between stud-
ies using waiting lists or placebo), whether effects had re-
mained constant in follow-up assessments, and how often indi-
cations of publication bias were found.
4. We compared the mean baseline severity in pharmacotherapy and
psychotherapy trials on major depressive disorder (MDD) in-
cluded in evaluations by Turner et al15
and Cuijpers et al.16Because
such an analysis requires a sufficient number of studies for both
treatment modalities, it was not possible for other disorders.
Nonparametric tests were used throughout our study. Group means
were compared with Mann-Whitney tests and dichotomous data
with χ2tests; the α level was set at P < .05. Because all analyses were
considered exploratory rather than confirmatory, adjustments for
multiple testing were not made.
Results
The search retrieved 45 233 responses; 20 703 remained after elimi-
nation of duplicates. Detailed PRISMA flowcharts on the selection pro-
cess are provided in the Supplement (eAppendix 5).9 We included 61
meta-analyses (mean AMSTAR score, 8.4 [95% CI, 7.8-9.0]) on 21 dis-
orders (852 individual trials and 137 126 participants). Thirty-three
(54.1%) meta-analyses examined pharmacotherapy15,17-48
; 17
(27.9%), psychotherapy16,19,49-63
(most focused on CBT or specific
treatments; very few examined psychodynamic approaches50,59
);
7 (11.5%), direct comparisons49,59,64-68
; and 12, combination
therapies (19.7%)49,57,59,65,68-75
(several meta-analyses included
>1 comparison).
Figure 1, Figure 2, and Figure 3 present the effect sizes in SMD
units. Because of the limitations of effect size estimates, we strongly
recommend that readers review the detailed narrative description
of the underlying meta-analyses, including the drug classes ana-
lyzed, responder rates, RRs, ARDs, NNTs, and SMDs in the Supple-
ment (eAppendix 6, with eTable 5). The sequence we used for our
report always follows the order of the disorders in the ICD-10.
Pharmacotherapy and Psychotherapy Compared With
Placebo or No Treatment
There were eligible reviews for at least one treatment modality for
all a priori–selected disorders except personality disorders other than
borderline personality disorder, impulse control disorders other than
trichotillomania, and substance-related disorders other than alco-
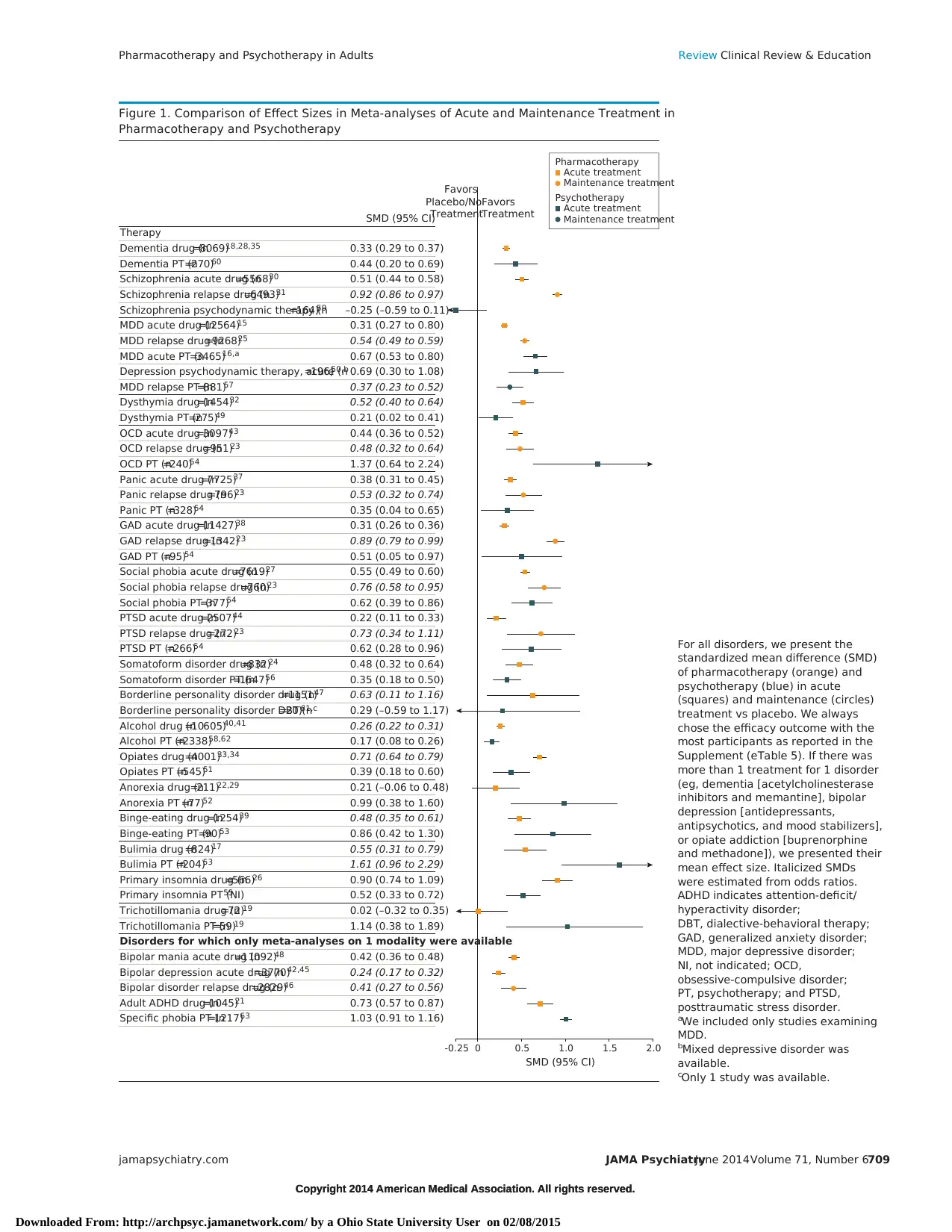
hol and opiate dependence (Figure 1). Most meta-analyses exam-
ined acute treatment; only 5 examined maintenance treatment. All
but 5 reviews demonstrated statistical significance compared with
placebo, which could be expected because only treatments recom-
mended by guidelines were chosen. However, the mean SMD of all
meta-analyses (0.50 [95% CI, 0.41-0.59]) suggests medium effi-
cacy of psychiatric treatments according to Cohen,8 and few treat-
ments had large effect sizes (mean SMD, ⱖ0.80).19,21,23,26,33,52-54,63
Acute-phase psychotherapy effect sizes for the same disorder tended
to be larger than those of pharmacotherapy (mean SMD, 0.40 [95%
CI, 0.28-0.52] vs 0.58 [0.40-0.76]). However, Figure 1 also shows
that the number of patients included in the acute-phase psycho-
therapy meta-analyses (median and mean sample sizes, 270 and 595,
respectively) was generally smaller than in the pharmacotherapy
studies (2507 and 3623) (U = 650.00; P < .001). The lower sample
sizes resulted in broader CIs and, thus, more uncertainty about the
true SMD. Finally, maintenance treatment with psychotropic drugs
was associated with consistently larger effect sizes than was acute
treatment.
Head-to-Head Pharmacotherapy and Psychotherapy
Seven meta-analyses, often with small sample sizes (range, 92-
1662; median 375), on schizophrenia,59 MDD,64,67 dysthymic
disorder,49 panic disorder,68 generalized anxiety disorder,66 social
phobia,68 and bulimia65compared pharmacotherapy and psycho-
therapy head-to-head. Although there was a trend in favor of psy-
chotherapy, this trend was significant only for relapse prevention in
depression64and for bulimia65; pharmacotherapy was more effec-
tive for dysthymic disorder49and schizophrenia (compared with psy-
chodynamic therapy)59(Figure 2).
Combinations of Pharmacotherapy and Psychotherapy
Twelve meta-analyses, also with small sample sizes (range, 23-
2131; median, 256) on schizophrenia,59,74MDD,70,71
dysthymic
disorder,49 bipolar disorder,57panic disorder,72,75
social phobia,68
posttraumatic stress disorder,73opiate addiction,69 and bulimia65
examined the effects of combining pharmacotherapy with psycho-
therapy.All analyses,except those on posttraumatic stress
Clinical Review & EducationReview Pharmacotherapy and Psychotherapy in Adults
708 JAMA PsychiatryJune 2014Volume 71, Number 6 jamapsychiatry.com
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
ity) and assessed their quality with the Cochrane risk of bias tool,13
which is based on scientific evidence of associations between over-
estimates of effect and methodologic shortcomings in the follow-
ing domains:
1. appropriate randomization (eg, computer-generated random-
ization sequence),
2. allocation concealment (eg, voice mail system),
3. blinding (because blinding of therapists is impossible in psycho-
therapy trials, we compared blind outcome assessment with no
blinding), and
4. missing outcomes (whether patients who withdrew from the
study and their reasons for withdrawal were reported, an inten-
tion-to-treat analysis was performed, and we also reported the
size of the overall dropout rate).
We did not examine the tool’s domain selective reporting but
added an item to the control groups that we classified as any con-
temporaneous treatment (placebo, treatment as usual, or ineffec-
tive therapy) vs wait list or no treatment. All items were rated as low,
unclear, and high risk of bias, with items rated as unclear (eg, indi-
cated as randomized without further explanation) combined with
those having a high risk of bias in the statistical analysis. Moreover,
we analyzed how often follow-up data after the trial end point were
collected.
Statistical Analysis
Statistical analysis was conducted in 4 steps.
1. We compared the sample sizes of psychotherapy and pharma-
cotherapy meta-analyses and their quality as measured by A Mea-
surement Tool to Assess Systematic Reviews (AMSTAR) score.14
2. We compared the sample sizes and the methodologic quality of
the individualacute-phase studies included in the psycho-
therapy and pharmacotherapy meta-analyses with the Coch-
rane risk of bias tool.13The same analysis was performed on the
single disorders (Supplement [eAppendix 4, eFigures 1-7]).
3. We examined whether the meta-analyses conducted subgroup
comparisons on the risk of bias tool domains (eg, between stud-
ies using waiting lists or placebo), whether effects had re-
mained constant in follow-up assessments, and how often indi-
cations of publication bias were found.
4. We compared the mean baseline severity in pharmacotherapy and
psychotherapy trials on major depressive disorder (MDD) in-
cluded in evaluations by Turner et al15
and Cuijpers et al.16Because
such an analysis requires a sufficient number of studies for both
treatment modalities, it was not possible for other disorders.
Nonparametric tests were used throughout our study. Group means
were compared with Mann-Whitney tests and dichotomous data
with χ2tests; the α level was set at P < .05. Because all analyses were
considered exploratory rather than confirmatory, adjustments for
multiple testing were not made.
Results
The search retrieved 45 233 responses; 20 703 remained after elimi-
nation of duplicates. Detailed PRISMA flowcharts on the selection pro-
cess are provided in the Supplement (eAppendix 5).9 We included 61
meta-analyses (mean AMSTAR score, 8.4 [95% CI, 7.8-9.0]) on 21 dis-
orders (852 individual trials and 137 126 participants). Thirty-three
(54.1%) meta-analyses examined pharmacotherapy15,17-48
; 17
(27.9%), psychotherapy16,19,49-63
(most focused on CBT or specific
treatments; very few examined psychodynamic approaches50,59
);
7 (11.5%), direct comparisons49,59,64-68
; and 12, combination
therapies (19.7%)49,57,59,65,68-75
(several meta-analyses included
>1 comparison).
Figure 1, Figure 2, and Figure 3 present the effect sizes in SMD
units. Because of the limitations of effect size estimates, we strongly
recommend that readers review the detailed narrative description
of the underlying meta-analyses, including the drug classes ana-
lyzed, responder rates, RRs, ARDs, NNTs, and SMDs in the Supple-
ment (eAppendix 6, with eTable 5). The sequence we used for our
report always follows the order of the disorders in the ICD-10.
Pharmacotherapy and Psychotherapy Compared With
Placebo or No Treatment
There were eligible reviews for at least one treatment modality for
all a priori–selected disorders except personality disorders other than
borderline personality disorder, impulse control disorders other than
trichotillomania, and substance-related disorders other than alco-
hol and opiate dependence (Figure 1). Most meta-analyses exam-
ined acute treatment; only 5 examined maintenance treatment. All
but 5 reviews demonstrated statistical significance compared with
placebo, which could be expected because only treatments recom-
mended by guidelines were chosen. However, the mean SMD of all
meta-analyses (0.50 [95% CI, 0.41-0.59]) suggests medium effi-
cacy of psychiatric treatments according to Cohen,8 and few treat-
ments had large effect sizes (mean SMD, ⱖ0.80).19,21,23,26,33,52-54,63
Acute-phase psychotherapy effect sizes for the same disorder tended
to be larger than those of pharmacotherapy (mean SMD, 0.40 [95%
CI, 0.28-0.52] vs 0.58 [0.40-0.76]). However, Figure 1 also shows
that the number of patients included in the acute-phase psycho-
therapy meta-analyses (median and mean sample sizes, 270 and 595,
respectively) was generally smaller than in the pharmacotherapy
studies (2507 and 3623) (U = 650.00; P < .001). The lower sample
sizes resulted in broader CIs and, thus, more uncertainty about the
true SMD. Finally, maintenance treatment with psychotropic drugs
was associated with consistently larger effect sizes than was acute
treatment.
Head-to-Head Pharmacotherapy and Psychotherapy
Seven meta-analyses, often with small sample sizes (range, 92-
1662; median 375), on schizophrenia,59 MDD,64,67 dysthymic
disorder,49 panic disorder,68 generalized anxiety disorder,66 social
phobia,68 and bulimia65compared pharmacotherapy and psycho-
therapy head-to-head. Although there was a trend in favor of psy-
chotherapy, this trend was significant only for relapse prevention in
depression64and for bulimia65; pharmacotherapy was more effec-
tive for dysthymic disorder49and schizophrenia (compared with psy-
chodynamic therapy)59(Figure 2).
Combinations of Pharmacotherapy and Psychotherapy
Twelve meta-analyses, also with small sample sizes (range, 23-
2131; median, 256) on schizophrenia,59,74MDD,70,71
dysthymic
disorder,49 bipolar disorder,57panic disorder,72,75
social phobia,68
posttraumatic stress disorder,73opiate addiction,69 and bulimia65
examined the effects of combining pharmacotherapy with psycho-
therapy.All analyses,except those on posttraumatic stress
Clinical Review & EducationReview Pharmacotherapy and Psychotherapy in Adults
708 JAMA PsychiatryJune 2014Volume 71, Number 6 jamapsychiatry.com
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide
Copyright 2014 American Medical Association. All rights reserved.
Figure 1. Comparison of Effect Sizes in Meta-analyses of Acute and Maintenance Treatment in
Pharmacotherapy and Psychotherapy
-0.25 1.0 2.00.5 1.5
SMD (95% CI)
0
Favors
Placebo/No
Treatment
Favors
Treatment
Therapy
SMD (95% CI)
0.33 (0.29 to 0.37)
0.44 (0.20 to 0.69)
0.51 (0.44 to 0.58)
0.92 (0.86 to 0.97)
–0.25 (–0.59 to 0.11)
0.31 (0.27 to 0.80)
0.54 (0.49 to 0.59)
0.67 (0.53 to 0.80)
0.69 (0.30 to 1.08)
0.37 (0.23 to 0.52)
0.52 (0.40 to 0.64)
0.21 (0.02 to 0.41)
0.44 (0.36 to 0.52)
0.48 (0.32 to 0.64)
1.37 (0.64 to 2.24)
0.38 (0.31 to 0.45)
0.53 (0.32 to 0.74)
0.35 (0.04 to 0.65)
0.31 (0.26 to 0.36)
0.89 (0.79 to 0.99)
0.51 (0.05 to 0.97)
0.55 (0.49 to 0.60)
0.76 (0.58 to 0.95)
0.62 (0.39 to 0.86)
0.22 (0.11 to 0.33)
0.73 (0.34 to 1.11)
0.62 (0.28 to 0.96)
0.48 (0.32 to 0.64)
0.35 (0.18 to 0.50)
0.63 (0.11 to 1.16)
0.29 (–0.59 to 1.17)
0.26 (0.22 to 0.31)
0.17 (0.08 to 0.26)
0.71 (0.64 to 0.79)
0.39 (0.18 to 0.60)
0.21 (–0.06 to 0.48)
0.99 (0.38 to 1.60)
0.48 (0.35 to 0.61)
0.86 (0.42 to 1.30)
0.55 (0.31 to 0.79)
1.61 (0.96 to 2.29)
0.90 (0.74 to 1.09)
0.52 (0.33 to 0.72)
0.02 (–0.32 to 0.35)
1.14 (0.38 to 1.89)
0.42 (0.36 to 0.48)
0.24 (0.17 to 0.32)
0.41 (0.27 to 0.56)
0.73 (0.57 to 0.87)
1.03 (0.91 to 1.16)
Dementia drug (n=8069)18,28,35
Dementia PT (n=270)60
Schizophrenia acute drug (n=5568)30
Schizophrenia relapse drug (n=6493)31
Schizophrenia psychodynamic therapy (n=164)59
MDD acute drug (n=12564)15
MDD relapse drug (n=9268)25
MDD acute PT (n=3465)16,a
Depression psychodynamic therapy, acute (n=196)50,b
MDD relapse PT (n=881)57
Dysthymia drug (n=1454)32
Dysthymia PT (n=275)49
OCD acute drug (n=3097)43
OCD relapse drug (n=951)23
OCD PT (n=240)54
Panic acute drug (n=7725)37
Panic relapse drug (n=796)23
Panic PT (n=328)54
GAD acute drug (n=11427)38
GAD relapse drug (n=1342)23
GAD PT (n=95)54
Social phobia acute drug (n=7619)27
Social phobia relapse drug (n=760)23
Social phobia PT (n=377)54
PTSD acute drug (n=2507)44
PTSD relapse drug (n=272)23
PTSD PT (n=266)54
Somatoform disorder drug (n=832)24
Somatoform disorder PT (n=1647)56
Borderline personality disorder drug (n=1151)47
Borderline personality disorder DBT (n=20)61,c
Alcohol drug (n=10605)40,41
Alcohol PT (n=2338)58,62
Opiates drug (n=4001)33,34
Opiates PT (n=545)51
Anorexia drug (n=211)22,29
Anorexia PT (n=77)52
Binge-eating drug (n=1254)39
Binge-eating PT (n=90)53
Bulimia drug (n=824)17
Bulimia PT (n=204)53
Primary insomnia drug (n=566)26
Primary insomnia PT (NI)55
Trichotillomania drug (n=72)19
Trichotillomania PT (n=59)19
Disorders for which only meta-analyses on 1 modality were available
Bipolar mania acute drug (n=11092)48
Bipolar depression acute drug (n=3770)42,45
Bipolar disorder relapse drug (n=2829)46
Adult ADHD drug (n=1045)21
Specific phobia PT (n=1217)63
Pharmacotherapy
Acute treatment
Maintenance treatment
Psychotherapy
Acute treatment
Maintenance treatment
For all disorders, we present the
standardized mean difference (SMD)
of pharmacotherapy (orange) and
psychotherapy (blue) in acute
(squares) and maintenance (circles)
treatment vs placebo. We always
chose the efficacy outcome with the
most participants as reported in the
Supplement (eTable 5). If there was
more than 1 treatment for 1 disorder
(eg, dementia [acetylcholinesterase
inhibitors and memantine], bipolar
depression [antidepressants,
antipsychotics, and mood stabilizers],
or opiate addiction [buprenorphine
and methadone]), we presented their
mean effect size. Italicized SMDs
were estimated from odds ratios.
ADHD indicates attention-deficit/
hyperactivity disorder;
DBT, dialective-behavioral therapy;
GAD, generalized anxiety disorder;
MDD, major depressive disorder;
NI, not indicated; OCD,
obsessive-compulsive disorder;
PT, psychotherapy; and PTSD,
posttraumatic stress disorder.
aWe included only studies examining
MDD.
bMixed depressive disorder was
available.
cOnly 1 study was available.
Pharmacotherapy and Psychotherapy in Adults Review Clinical Review & Education
jamapsychiatry.com JAMA PsychiatryJune 2014Volume 71, Number 6709
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
Figure 1. Comparison of Effect Sizes in Meta-analyses of Acute and Maintenance Treatment in
Pharmacotherapy and Psychotherapy
-0.25 1.0 2.00.5 1.5
SMD (95% CI)
0
Favors
Placebo/No
Treatment
Favors
Treatment
Therapy
SMD (95% CI)
0.33 (0.29 to 0.37)
0.44 (0.20 to 0.69)
0.51 (0.44 to 0.58)
0.92 (0.86 to 0.97)
–0.25 (–0.59 to 0.11)
0.31 (0.27 to 0.80)
0.54 (0.49 to 0.59)
0.67 (0.53 to 0.80)
0.69 (0.30 to 1.08)
0.37 (0.23 to 0.52)
0.52 (0.40 to 0.64)
0.21 (0.02 to 0.41)
0.44 (0.36 to 0.52)
0.48 (0.32 to 0.64)
1.37 (0.64 to 2.24)
0.38 (0.31 to 0.45)
0.53 (0.32 to 0.74)
0.35 (0.04 to 0.65)
0.31 (0.26 to 0.36)
0.89 (0.79 to 0.99)
0.51 (0.05 to 0.97)
0.55 (0.49 to 0.60)
0.76 (0.58 to 0.95)
0.62 (0.39 to 0.86)
0.22 (0.11 to 0.33)
0.73 (0.34 to 1.11)
0.62 (0.28 to 0.96)
0.48 (0.32 to 0.64)
0.35 (0.18 to 0.50)
0.63 (0.11 to 1.16)
0.29 (–0.59 to 1.17)
0.26 (0.22 to 0.31)
0.17 (0.08 to 0.26)
0.71 (0.64 to 0.79)
0.39 (0.18 to 0.60)
0.21 (–0.06 to 0.48)
0.99 (0.38 to 1.60)
0.48 (0.35 to 0.61)
0.86 (0.42 to 1.30)
0.55 (0.31 to 0.79)
1.61 (0.96 to 2.29)
0.90 (0.74 to 1.09)
0.52 (0.33 to 0.72)
0.02 (–0.32 to 0.35)
1.14 (0.38 to 1.89)
0.42 (0.36 to 0.48)
0.24 (0.17 to 0.32)
0.41 (0.27 to 0.56)
0.73 (0.57 to 0.87)
1.03 (0.91 to 1.16)
Dementia drug (n=8069)18,28,35
Dementia PT (n=270)60
Schizophrenia acute drug (n=5568)30
Schizophrenia relapse drug (n=6493)31
Schizophrenia psychodynamic therapy (n=164)59
MDD acute drug (n=12564)15
MDD relapse drug (n=9268)25
MDD acute PT (n=3465)16,a
Depression psychodynamic therapy, acute (n=196)50,b
MDD relapse PT (n=881)57
Dysthymia drug (n=1454)32
Dysthymia PT (n=275)49
OCD acute drug (n=3097)43
OCD relapse drug (n=951)23
OCD PT (n=240)54
Panic acute drug (n=7725)37
Panic relapse drug (n=796)23
Panic PT (n=328)54
GAD acute drug (n=11427)38
GAD relapse drug (n=1342)23
GAD PT (n=95)54
Social phobia acute drug (n=7619)27
Social phobia relapse drug (n=760)23
Social phobia PT (n=377)54
PTSD acute drug (n=2507)44
PTSD relapse drug (n=272)23
PTSD PT (n=266)54
Somatoform disorder drug (n=832)24
Somatoform disorder PT (n=1647)56
Borderline personality disorder drug (n=1151)47
Borderline personality disorder DBT (n=20)61,c
Alcohol drug (n=10605)40,41
Alcohol PT (n=2338)58,62
Opiates drug (n=4001)33,34
Opiates PT (n=545)51
Anorexia drug (n=211)22,29
Anorexia PT (n=77)52
Binge-eating drug (n=1254)39
Binge-eating PT (n=90)53
Bulimia drug (n=824)17
Bulimia PT (n=204)53
Primary insomnia drug (n=566)26
Primary insomnia PT (NI)55
Trichotillomania drug (n=72)19
Trichotillomania PT (n=59)19
Disorders for which only meta-analyses on 1 modality were available
Bipolar mania acute drug (n=11092)48
Bipolar depression acute drug (n=3770)42,45
Bipolar disorder relapse drug (n=2829)46
Adult ADHD drug (n=1045)21
Specific phobia PT (n=1217)63
Pharmacotherapy
Acute treatment
Maintenance treatment
Psychotherapy
Acute treatment
Maintenance treatment
For all disorders, we present the
standardized mean difference (SMD)
of pharmacotherapy (orange) and
psychotherapy (blue) in acute
(squares) and maintenance (circles)
treatment vs placebo. We always
chose the efficacy outcome with the
most participants as reported in the
Supplement (eTable 5). If there was
more than 1 treatment for 1 disorder
(eg, dementia [acetylcholinesterase
inhibitors and memantine], bipolar
depression [antidepressants,
antipsychotics, and mood stabilizers],
or opiate addiction [buprenorphine
and methadone]), we presented their
mean effect size. Italicized SMDs
were estimated from odds ratios.
ADHD indicates attention-deficit/
hyperactivity disorder;
DBT, dialective-behavioral therapy;
GAD, generalized anxiety disorder;
MDD, major depressive disorder;
NI, not indicated; OCD,
obsessive-compulsive disorder;
PT, psychotherapy; and PTSD,
posttraumatic stress disorder.
aWe included only studies examining
MDD.
bMixed depressive disorder was
available.
cOnly 1 study was available.
Pharmacotherapy and Psychotherapy in Adults Review Clinical Review & Education
jamapsychiatry.com JAMA PsychiatryJune 2014Volume 71, Number 6709
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

Copyright 2014 American Medical Association. All rights reserved.
disorder73
and psychodynamic therapy for schizophrenia,59showed
a trend in favor of combination therapy, which was statistically sig-
nificant in 7 studies. The combination was better when psycho-
therapy was added to drug therapy and vice versa (Figure 3).
Comparison of the Quality of Research
Quality of Pharmacotherapy and Psychotherapy Meta-analyses
No significant difference exists in the overall quality of pharmaco-
therapy and psychotherapy meta-analyses based on AMSTAR total
scores (mean, 8.7 [95% CI, 7.9-9.4] vs 8.2 [7.1-9.3]; Mann-Whitney
test, U = 277.500; P = .38) or subscores apart from conflict of in-
terest, which was reported by all pharmacotherapy meta-analyses
but only by 63% of psychotherapy meta-analyses (χ2
1 = 12.55;
P < .001) (Supplement [eAppendix 4, eTable 3]).
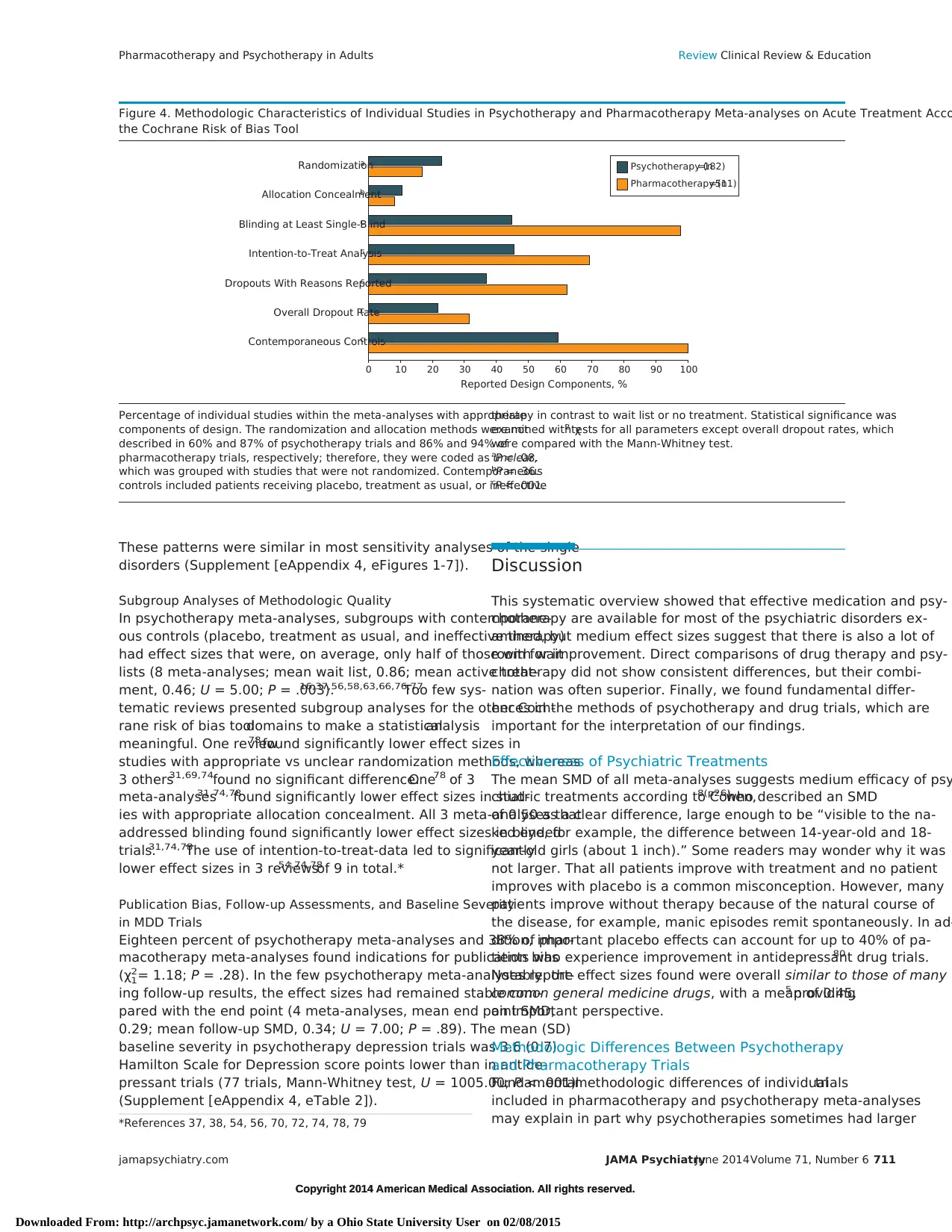
Quality of the Individual Included Trials
Only acute-phase studies with overall 113 833 participants were in-
cluded to enhance comparability. The median and mean sizes of the
182 individual psychotherapy trials were significantly lower (62 and
89) than those of the 511 individual pharmacotherapy trials (117 and
191) (U = 63 036; P < .001).
More individual pharmacotherapy vs psychotherapy trials used
at least blind outcome assessment (97.7% vs 44.5%; χ2
1= 281.92;
P < .001), used placebo controls rather than wait lists (99.2% vs
59.3%; χ2
1= 213.66; P < .001), used intention-to-treat data (68.7%
vs 45.1%; χ2
1= 31.98; P < .001), and reported withdrawal rates with
their reasons (62.2% vs 36.3%; χ2
1= 38.64; P < .001).13
In contrast,
psychotherapy trials had overall lower withdrawal rates (31.0% vs
21.3%; U = 48 217; P < .001).
Most trials of both modalities were described as randomized
without any description of the methods (appropriate randomiza-
tion in psychotherapy vs pharmacotherapy, 22.5% vs 16.8%;
χ 2
1= 2.91; P = .08; appropriate allocation concealment, 10.4% vs
8.2%; χ2
1= 0.82; P = .36) (Figure 4).
Only 4.9% of pharmacotherapy trials provided follow-up data
compared with 54.9% of psychotherapy trials (χ2
1= 227.42; P < .001).
Figure 3. Comparison of Effect Sizes in Meta-analyses of Alone vs Combined Pharmacotherapy
or Psychotherapy
-0.5 0.5 1.00
SMD (95% CI)
Favors
Monotherapy
Favors Combination
TherapyDiagnosis SMD (95% CI)
Schizophrenia PDT (n=24)59 0.00 (-0.86 to 0.86)
Major depressive disorder (n=903)71 0.28 (0.13 to 0.43)
Panic disorder (n=532)72,75,a 0.17 (-0.03 to 0.36)
Social phobia (n=267)68 0.42 (0.18 to 0.68)
Posttraumatic stress disorder (n=65)73 0.38 (-0.11 to 0.87)
Bulimia (n=257)65 0.29 (0.02 to 0.86)
Pharmacotherapy added to psychotherapy
Schizophrenia CBT (n=2131)74 0.33 (0.21 to 0.45)
Schizophrenia PDT (n=90)59 0.07 (-0.34 to 0.49)
Depressive disorder (n=2036)70 0.31 (0.20 to 0.40)
Dysthymic disorder (n=541)49 0.04 (-0.17 to 0.24)
Bipolar disorder (n=487)57 0.19 (-0.21 to 0.60)
Panic disorder (n=746)72,75 0.24 (0.08 to 0.40)
Psychotherapy added to pharmacotherapy
Social phobia (n=256)68 0.12 (-0.13 to 0.36)
Posttraumatic stress disorder (n=23)73 -0.33 (-1.17 to 0.51)
Bulimia (n=141)65 0.49 (0.09 to 0.89)
Opioid dependence (n=232)69 0.12 (0.45 to -0.21)
The outcome in all studies was
reduction of overall symptoms.
Italicized standardized mean
differences (SMDs) were estimated
from odds ratios. CBT indicates
cognitive-behavioral therapy;
PDT, psychodynamic therapy.
aFor panic disorder, the effect size is
the mean of 2 meta-analyses.72,75
Figure 2. Comparison of Effect Sizes in Meta-analyses of Direct Comparisons of Pharmacotherapy
and Psychotherapy
-1.00 0.50 1.000.00
SMD (95% CI)
-0.50
Favors
Drug
Favors
PsychotherapyDiagnosis SMD (95% CI)
Schizophrenia (n=92)59,a -0.56 (-0.98 to -0.14)
Major depressive disorder, acute (n=1662)67 -0.05 (-0.24 to 0.13)
Major depressive disorder, relapse (n=231)64 -0.71 (-0.40 to 1.01)
Dysthymic disorder (n=874)49 -0.47 (-0.75 to -0.18)
Panic disorder (n=375)68 0.08 (-0.13 to 0.28)
Generalized anxiety disorder (NI)66 0.33 (-0.02 to 0.67)
Social phobia (n=208)68 0.15 (-0.12 to 0.43)
Bulimia (n=237)65 0.52 (0.20 to 0.84)
The outcome in all studies was
reduction of overall symptoms except
for relapse prevention in depression.
Italicized standardized mean
differences (SMDs) were estimated
from odds ratios. NI indicates not
indicated.
aDrug vs psychodynamic
psychotherapy.
Clinical Review & EducationReview Pharmacotherapy and Psychotherapy in Adults
710 JAMA PsychiatryJune 2014Volume 71, Number 6 jamapsychiatry.com
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
disorder73
and psychodynamic therapy for schizophrenia,59showed
a trend in favor of combination therapy, which was statistically sig-
nificant in 7 studies. The combination was better when psycho-
therapy was added to drug therapy and vice versa (Figure 3).
Comparison of the Quality of Research
Quality of Pharmacotherapy and Psychotherapy Meta-analyses
No significant difference exists in the overall quality of pharmaco-
therapy and psychotherapy meta-analyses based on AMSTAR total
scores (mean, 8.7 [95% CI, 7.9-9.4] vs 8.2 [7.1-9.3]; Mann-Whitney
test, U = 277.500; P = .38) or subscores apart from conflict of in-
terest, which was reported by all pharmacotherapy meta-analyses
but only by 63% of psychotherapy meta-analyses (χ2
1 = 12.55;
P < .001) (Supplement [eAppendix 4, eTable 3]).
Quality of the Individual Included Trials
Only acute-phase studies with overall 113 833 participants were in-
cluded to enhance comparability. The median and mean sizes of the
182 individual psychotherapy trials were significantly lower (62 and
89) than those of the 511 individual pharmacotherapy trials (117 and
191) (U = 63 036; P < .001).
More individual pharmacotherapy vs psychotherapy trials used
at least blind outcome assessment (97.7% vs 44.5%; χ2
1= 281.92;
P < .001), used placebo controls rather than wait lists (99.2% vs
59.3%; χ2
1= 213.66; P < .001), used intention-to-treat data (68.7%
vs 45.1%; χ2
1= 31.98; P < .001), and reported withdrawal rates with
their reasons (62.2% vs 36.3%; χ2
1= 38.64; P < .001).13
In contrast,
psychotherapy trials had overall lower withdrawal rates (31.0% vs
21.3%; U = 48 217; P < .001).
Most trials of both modalities were described as randomized
without any description of the methods (appropriate randomiza-
tion in psychotherapy vs pharmacotherapy, 22.5% vs 16.8%;
χ 2
1= 2.91; P = .08; appropriate allocation concealment, 10.4% vs
8.2%; χ2
1= 0.82; P = .36) (Figure 4).
Only 4.9% of pharmacotherapy trials provided follow-up data
compared with 54.9% of psychotherapy trials (χ2
1= 227.42; P < .001).
Figure 3. Comparison of Effect Sizes in Meta-analyses of Alone vs Combined Pharmacotherapy
or Psychotherapy
-0.5 0.5 1.00
SMD (95% CI)
Favors
Monotherapy
Favors Combination
TherapyDiagnosis SMD (95% CI)
Schizophrenia PDT (n=24)59 0.00 (-0.86 to 0.86)
Major depressive disorder (n=903)71 0.28 (0.13 to 0.43)
Panic disorder (n=532)72,75,a 0.17 (-0.03 to 0.36)
Social phobia (n=267)68 0.42 (0.18 to 0.68)
Posttraumatic stress disorder (n=65)73 0.38 (-0.11 to 0.87)
Bulimia (n=257)65 0.29 (0.02 to 0.86)
Pharmacotherapy added to psychotherapy
Schizophrenia CBT (n=2131)74 0.33 (0.21 to 0.45)
Schizophrenia PDT (n=90)59 0.07 (-0.34 to 0.49)
Depressive disorder (n=2036)70 0.31 (0.20 to 0.40)
Dysthymic disorder (n=541)49 0.04 (-0.17 to 0.24)
Bipolar disorder (n=487)57 0.19 (-0.21 to 0.60)
Panic disorder (n=746)72,75 0.24 (0.08 to 0.40)
Psychotherapy added to pharmacotherapy
Social phobia (n=256)68 0.12 (-0.13 to 0.36)
Posttraumatic stress disorder (n=23)73 -0.33 (-1.17 to 0.51)
Bulimia (n=141)65 0.49 (0.09 to 0.89)
Opioid dependence (n=232)69 0.12 (0.45 to -0.21)
The outcome in all studies was
reduction of overall symptoms.
Italicized standardized mean
differences (SMDs) were estimated
from odds ratios. CBT indicates
cognitive-behavioral therapy;
PDT, psychodynamic therapy.
aFor panic disorder, the effect size is
the mean of 2 meta-analyses.72,75
Figure 2. Comparison of Effect Sizes in Meta-analyses of Direct Comparisons of Pharmacotherapy
and Psychotherapy
-1.00 0.50 1.000.00
SMD (95% CI)
-0.50
Favors
Drug
Favors
PsychotherapyDiagnosis SMD (95% CI)
Schizophrenia (n=92)59,a -0.56 (-0.98 to -0.14)
Major depressive disorder, acute (n=1662)67 -0.05 (-0.24 to 0.13)
Major depressive disorder, relapse (n=231)64 -0.71 (-0.40 to 1.01)
Dysthymic disorder (n=874)49 -0.47 (-0.75 to -0.18)
Panic disorder (n=375)68 0.08 (-0.13 to 0.28)
Generalized anxiety disorder (NI)66 0.33 (-0.02 to 0.67)
Social phobia (n=208)68 0.15 (-0.12 to 0.43)
Bulimia (n=237)65 0.52 (0.20 to 0.84)
The outcome in all studies was
reduction of overall symptoms except
for relapse prevention in depression.
Italicized standardized mean
differences (SMDs) were estimated
from odds ratios. NI indicates not
indicated.
aDrug vs psychodynamic
psychotherapy.
Clinical Review & EducationReview Pharmacotherapy and Psychotherapy in Adults
710 JAMA PsychiatryJune 2014Volume 71, Number 6 jamapsychiatry.com
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
Copyright 2014 American Medical Association. All rights reserved.
These patterns were similar in most sensitivity analyses of the single
disorders (Supplement [eAppendix 4, eFigures 1-7]).
Subgroup Analyses of Methodologic Quality
In psychotherapy meta-analyses, subgroups with contemporane-
ous controls (placebo, treatment as usual, and ineffective therapy)
had effect sizes that were, on average, only half of those with wait
lists (8 meta-analyses; mean wait list, 0.86; mean active treat-
ment, 0.46; U = 5.00; P = .003).16,37,56,58,63,66,76,77
Too few sys-
tematic reviews presented subgroup analyses for the other Coch-
rane risk of bias tooldomains to make a statisticalanalysis
meaningful. One review78 found significantly lower effect sizes in
studies with appropriate vs unclear randomization methods, whereas
3 others31,69,74found no significant difference.One78 of 3
meta-analyses31,74,78
found significantly lower effect sizes in stud-
ies with appropriate allocation concealment. All 3 meta-analyses that
addressed blinding found significantly lower effect sizes in blinded
trials.31,74,78
The use of intention-to-treat-data led to significantly
lower effect sizes in 3 reviews54,74,78
of 9 in total.*
Publication Bias, Follow-up Assessments, and Baseline Severity
in MDD Trials
Eighteen percent of psychotherapy meta-analyses and 38% of phar-
macotherapy meta-analyses found indications for publication bias
(χ2
1= 1.18; P = .28). In the few psychotherapy meta-analyses report-
ing follow-up results, the effect sizes had remained stable com-
pared with the end point (4 meta-analyses, mean end point SMD,
0.29; mean follow-up SMD, 0.34; U = 7.00; P = .89). The mean (SD)
baseline severity in psychotherapy depression trials was 3.6 (0.7)
Hamilton Scale for Depression score points lower than in antide-
pressant trials (77 trials, Mann-Whitney test, U = 1005.00; P < .001)
(Supplement [eAppendix 4, eTable 2]).
Discussion
This systematic overview showed that effective medication and psy-
chotherapy are available for most of the psychiatric disorders ex-
amined, but medium effect sizes suggest that there is also a lot of
room for improvement. Direct comparisons of drug therapy and psy-
chotherapy did not show consistent differences, but their combi-
nation was often superior. Finally, we found fundamental differ-
ences in the methods of psychotherapy and drug trials, which are
important for the interpretation of our findings.
Effectiveness of Psychiatric Treatments
The mean SMD of all meta-analyses suggests medium efficacy of psy
chiatric treatments according to Cohen,8(p26)
who described an SMD
of 0.50 as a clear difference, large enough to be “visible to the na-
ked eye, for example, the difference between 14-year-old and 18-
year-old girls (about 1 inch).” Some readers may wonder why it was
not larger. That all patients improve with treatment and no patient
improves with placebo is a common misconception. However, many
patients improve without therapy because of the natural course of
the disease, for example, manic episodes remit spontaneously. In ad-
dition, important placebo effects can account for up to 40% of pa-
tients who experience improvement in antidepressant drug trials.80
Notably, the effect sizes found were overall similar to those of many
common general medicine drugs, with a mean of 0.45,5 providing
an important perspective.
Methodologic Differences Between Psychotherapy
and Pharmacotherapy Trials
Fundamentalmethodologic differences of individualtrials
included in pharmacotherapy and psychotherapy meta-analyses
may explain in part why psychotherapies sometimes had larger*References 37, 38, 54, 56, 70, 72, 74, 78, 79
Figure 4. Methodologic Characteristics of Individual Studies in Psychotherapy and Pharmacotherapy Meta-analyses on Acute Treatment Acco
the Cochrane Risk of Bias Tool
Contemporaneous Controlsc
Overall Dropout Ratec
Dropouts With Reasons Reportedc
Intention-to-Treat Analysisc
Blinding at Least Single-Blindc
Allocation Concealmentb
Randomizationa
0 10 20 30 40 50 60 70 80 90 100
Reported Design Components, %
Psychotherapy (n=182)
Pharmacotherapy (n=511)
Percentage of individual studies within the meta-analyses with appropriate
components of design. The randomization and allocation methods were not
described in 60% and 87% of psychotherapy trials and 86% and 94% of
pharmacotherapy trials, respectively; therefore, they were coded as unclear,
which was grouped with studies that were not randomized. Contemporaneous
controls included patients receiving placebo, treatment as usual, or ineffective
therapy in contrast to wait list or no treatment. Statistical significance was
examined with χ2 tests for all parameters except overall dropout rates, which
were compared with the Mann-Whitney test.
aP = .08.
bP = .36.
cP < .001.
Pharmacotherapy and Psychotherapy in Adults Review Clinical Review & Education
jamapsychiatry.com JAMA PsychiatryJune 2014Volume 71, Number 6 711
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
These patterns were similar in most sensitivity analyses of the single
disorders (Supplement [eAppendix 4, eFigures 1-7]).
Subgroup Analyses of Methodologic Quality
In psychotherapy meta-analyses, subgroups with contemporane-
ous controls (placebo, treatment as usual, and ineffective therapy)
had effect sizes that were, on average, only half of those with wait
lists (8 meta-analyses; mean wait list, 0.86; mean active treat-
ment, 0.46; U = 5.00; P = .003).16,37,56,58,63,66,76,77
Too few sys-
tematic reviews presented subgroup analyses for the other Coch-
rane risk of bias tooldomains to make a statisticalanalysis
meaningful. One review78 found significantly lower effect sizes in
studies with appropriate vs unclear randomization methods, whereas
3 others31,69,74found no significant difference.One78 of 3
meta-analyses31,74,78
found significantly lower effect sizes in stud-
ies with appropriate allocation concealment. All 3 meta-analyses that
addressed blinding found significantly lower effect sizes in blinded
trials.31,74,78
The use of intention-to-treat-data led to significantly
lower effect sizes in 3 reviews54,74,78
of 9 in total.*
Publication Bias, Follow-up Assessments, and Baseline Severity
in MDD Trials
Eighteen percent of psychotherapy meta-analyses and 38% of phar-
macotherapy meta-analyses found indications for publication bias
(χ2
1= 1.18; P = .28). In the few psychotherapy meta-analyses report-
ing follow-up results, the effect sizes had remained stable com-
pared with the end point (4 meta-analyses, mean end point SMD,
0.29; mean follow-up SMD, 0.34; U = 7.00; P = .89). The mean (SD)
baseline severity in psychotherapy depression trials was 3.6 (0.7)
Hamilton Scale for Depression score points lower than in antide-
pressant trials (77 trials, Mann-Whitney test, U = 1005.00; P < .001)
(Supplement [eAppendix 4, eTable 2]).
Discussion
This systematic overview showed that effective medication and psy-
chotherapy are available for most of the psychiatric disorders ex-
amined, but medium effect sizes suggest that there is also a lot of
room for improvement. Direct comparisons of drug therapy and psy-
chotherapy did not show consistent differences, but their combi-
nation was often superior. Finally, we found fundamental differ-
ences in the methods of psychotherapy and drug trials, which are
important for the interpretation of our findings.
Effectiveness of Psychiatric Treatments
The mean SMD of all meta-analyses suggests medium efficacy of psy
chiatric treatments according to Cohen,8(p26)
who described an SMD
of 0.50 as a clear difference, large enough to be “visible to the na-
ked eye, for example, the difference between 14-year-old and 18-
year-old girls (about 1 inch).” Some readers may wonder why it was
not larger. That all patients improve with treatment and no patient
improves with placebo is a common misconception. However, many
patients improve without therapy because of the natural course of
the disease, for example, manic episodes remit spontaneously. In ad-
dition, important placebo effects can account for up to 40% of pa-
tients who experience improvement in antidepressant drug trials.80
Notably, the effect sizes found were overall similar to those of many
common general medicine drugs, with a mean of 0.45,5 providing
an important perspective.
Methodologic Differences Between Psychotherapy
and Pharmacotherapy Trials
Fundamentalmethodologic differences of individualtrials
included in pharmacotherapy and psychotherapy meta-analyses
may explain in part why psychotherapies sometimes had larger*References 37, 38, 54, 56, 70, 72, 74, 78, 79
Figure 4. Methodologic Characteristics of Individual Studies in Psychotherapy and Pharmacotherapy Meta-analyses on Acute Treatment Acco
the Cochrane Risk of Bias Tool
Contemporaneous Controlsc
Overall Dropout Ratec
Dropouts With Reasons Reportedc
Intention-to-Treat Analysisc
Blinding at Least Single-Blindc
Allocation Concealmentb
Randomizationa
0 10 20 30 40 50 60 70 80 90 100
Reported Design Components, %
Psychotherapy (n=182)
Pharmacotherapy (n=511)
Percentage of individual studies within the meta-analyses with appropriate
components of design. The randomization and allocation methods were not
described in 60% and 87% of psychotherapy trials and 86% and 94% of
pharmacotherapy trials, respectively; therefore, they were coded as unclear,
which was grouped with studies that were not randomized. Contemporaneous
controls included patients receiving placebo, treatment as usual, or ineffective
therapy in contrast to wait list or no treatment. Statistical significance was
examined with χ2 tests for all parameters except overall dropout rates, which
were compared with the Mann-Whitney test.
aP = .08.
bP = .36.
cP < .001.
Pharmacotherapy and Psychotherapy in Adults Review Clinical Review & Education
jamapsychiatry.com JAMA PsychiatryJune 2014Volume 71, Number 6 711
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide

Copyright 2014 American Medical Association. All rights reserved.
effect sizes vs control conditions compared with medication (eg,
MDD, obsessive-compulsive disorder, posttraumatic stress disor-
der, and bulimia; the opposite also exists, eg, methadone for opiate
addiction).16,33,53,54
However, direct comparisons produced no con-
sistent differences (eg, SMD for psychotherapy vs placebo or no
treatment for MDD, 0.67; antidepressants vs placebo, 0.31; but an-
tidepressants vs psychotherapy, 0.05).
Trikalinos et al81showed that systematic reviews in psychia-
try with participant numbers below 1000 do not provide robust
results. Small-study bias in individual psychotherapy trials and
meta-analyses, which were an order of magnitude smaller than
their pharmacotherapeutic counterparts, may have led to greater
effect sizes.82 For example, patients in small psychotherapy stud-
ies may be well selected because psychotherapy requires active
participation. In contrast, participants in multicenter drug trials
are often recruited by advertisements, attracting so-called “pro-
fessional patients.” Similarly, therapists in psychotherapy trials are
often well-trained experts. Researcher allegiance means that the
testing of a psychotherapy by its inventors might positively influ-
ence the effect size.83 Fewer psychotherapy than pharmaco-
therapy meta-analyses indicated their authors’ conflicts of inter-
est, although conflicts are not restricted to financial ones. These
factors may lead to higher generalizability in pharmacotherapy
trials in exchange for more “statisticalnoise” and lower effect
sizes. Obviously, the difference in sample size also results from
imbalances in funding when powerful pharmaceutical companies
sponsor drug trials.More public funding for psychotherapy
research is needed.
Psychotherapists always know whether they deliver active or
placebo treatment, and patients may realize whether they receive
“real” psychotherapy or not. However, even blind outcome assess-
ment was applied in only approximately 45% of psychotherapy trials
in contrast to 98% in drug trials. The few meta-analyses that ad-
dressed blinding identified bias.31,74,78
For example, blinded psy-
chotherapy trials for depression had significantly lower effect sizes,
and low-quality psychotherapy trials in general had a higher effect
size (SMD = 0.74) than high-quality trials (SMD = 0.22) (P < .001).78
In drug trials imperfect blinding can also be an issue if adverse ef-
fects allow one to guess the treatment group; unfortunately, this is
rarely tested.84
Nearly all individual drug trials used pill-placebo control groups;
41% of psychotherapy trials used wait-list controls, which almost
doubled effect sizes. Patients know they are waiting for therapy and
do not expect improvement. There can even be a “nocebo effect”
because being on a wait list can be a message of “you cannot im-
prove now.” In contrast, expectancy effects are considerable in pa-
tients receiving a placebo.
More than half of psychotherapy trials and a third of pharma-
cotherapy trials did not use an intention-to-treat-analysis. Per-
protocol analysis breaks randomization in that characteristics of
study completers in intervention and control groups can differ, for
example, because participants in placebo groups may withdraw pri-
marily because of inefficacy and patients in drug groups may with-
draw more because of adverse effects.85
Effect sizes for relapse prevention were generally larger than for
acute therapy with the same drugs that are given to a mixture of re-
sponders and nonresponders, lending strong support for continu-
ation treatment, but long-term treatment might sensitize brain re-
ceptors, facilitating relapse after abrupt withdrawal.86Psychotherapy
offers a more optimistic perspective because it could cure by induc-
ing lasting cognitive and behavioral changes. Indeed, the few psy-
chotherapy meta-analyses that examined follow-up after the study
ended found that treatment effects persisted.
Evidence for publication bias was numerically more frequent
in pharmacotherapy meta-analyses, where it is a well-known
phenomenon,1 5although it also occurs in psychotherapy
research.16Because the main driver for publication bias is trial fail-
ure and blind trials are more likely to fail, the infrequent use of
blinding in psychotherapy trials is important. Finally, the mean
baseline severity in psychotherapy trials on MDD was lower than
that in antidepressant trials. This could mean that psychotherapy
is particularly efficacious in milder forms of MDD in which antide-
pressants are less effective.4
Strengths and Limitations
In our study, the criteria of a systematic review were met, but obvi-
ously there are multiple limitations. Because the selection of the
meta-analyses was most prone to bias, we described this process
in detail (Supplement [eAppendix 5]). We always compared phar-
macotherapy and psychotherapy for the same disorders, and we
strongly discourage comparisons of treatments across disorders. Still,
meta-analyses depend on the quality of the included studies. To be
systematic, we chose the most recent meta-analyses because older
ones, although sometimes methodologically better, would have been
out of date. We analyzed classes of drugs rather than single agents,
assuming that the original authors had made an appropriate deci-
sion to pooldrugs. Therefore, our results are not specific, al-
though, with few exceptions, no large efficacy differences exist within
drug classes. We preferred psychotherapies under the umbrellas of
CBT and psychodynamic therapy; occasionally, more efficacious psy-
chotherapies may have been missed. We also did not examine ef-
fective interventions such as psychoeducation.87,88
To examine ad-
verse effects and balance them with efficacy was impossible, but this
domain is a major advantage of psychotherapy. Although based on
RCTs, meta-analysis itself is an observational method with many limi-
tations (eg, “garbage in garbage out” and heterogeneity), and RCTs
lack generalizability. Finally, the interpretation of effect size is con-
troversial. Therefore, we emphasize the importance of reading the
actual numbers in the Supplement (eAppendix 6).89However, meta-
analysis served as a useful tool to systematically provide a rough over-
view of what benefit psychiatry can offer patients. Even if other au-
thors made different choices, the chance of them reaching essentially
different conclusions is unlikely.
Conclusions
Because of the multiple methodologic differences between phar-
macotherapy and psychotherapy trials, indirect comparisons are
problematic. Meta-analysts should consistently address quality
indicators, such as control groups or degree of blinding. Head-to-
head trials are scarce, and funding for them is urgently needed.
Research should focus on how pharmacotherapy and psycho-
therapy can best be combined to obtain maximally synergistic
effects rather than debate the use of one treatment over the
other.
Clinical Review & EducationReview Pharmacotherapy and Psychotherapy in Adults
712 JAMA PsychiatryJune 2014Volume 71, Number 6 jamapsychiatry.com
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
effect sizes vs control conditions compared with medication (eg,
MDD, obsessive-compulsive disorder, posttraumatic stress disor-
der, and bulimia; the opposite also exists, eg, methadone for opiate
addiction).16,33,53,54
However, direct comparisons produced no con-
sistent differences (eg, SMD for psychotherapy vs placebo or no
treatment for MDD, 0.67; antidepressants vs placebo, 0.31; but an-
tidepressants vs psychotherapy, 0.05).
Trikalinos et al81showed that systematic reviews in psychia-
try with participant numbers below 1000 do not provide robust
results. Small-study bias in individual psychotherapy trials and
meta-analyses, which were an order of magnitude smaller than
their pharmacotherapeutic counterparts, may have led to greater
effect sizes.82 For example, patients in small psychotherapy stud-
ies may be well selected because psychotherapy requires active
participation. In contrast, participants in multicenter drug trials
are often recruited by advertisements, attracting so-called “pro-
fessional patients.” Similarly, therapists in psychotherapy trials are
often well-trained experts. Researcher allegiance means that the
testing of a psychotherapy by its inventors might positively influ-
ence the effect size.83 Fewer psychotherapy than pharmaco-
therapy meta-analyses indicated their authors’ conflicts of inter-
est, although conflicts are not restricted to financial ones. These
factors may lead to higher generalizability in pharmacotherapy
trials in exchange for more “statisticalnoise” and lower effect
sizes. Obviously, the difference in sample size also results from
imbalances in funding when powerful pharmaceutical companies
sponsor drug trials.More public funding for psychotherapy
research is needed.
Psychotherapists always know whether they deliver active or
placebo treatment, and patients may realize whether they receive
“real” psychotherapy or not. However, even blind outcome assess-
ment was applied in only approximately 45% of psychotherapy trials
in contrast to 98% in drug trials. The few meta-analyses that ad-
dressed blinding identified bias.31,74,78
For example, blinded psy-
chotherapy trials for depression had significantly lower effect sizes,
and low-quality psychotherapy trials in general had a higher effect
size (SMD = 0.74) than high-quality trials (SMD = 0.22) (P < .001).78
In drug trials imperfect blinding can also be an issue if adverse ef-
fects allow one to guess the treatment group; unfortunately, this is
rarely tested.84
Nearly all individual drug trials used pill-placebo control groups;
41% of psychotherapy trials used wait-list controls, which almost
doubled effect sizes. Patients know they are waiting for therapy and
do not expect improvement. There can even be a “nocebo effect”
because being on a wait list can be a message of “you cannot im-
prove now.” In contrast, expectancy effects are considerable in pa-
tients receiving a placebo.
More than half of psychotherapy trials and a third of pharma-
cotherapy trials did not use an intention-to-treat-analysis. Per-
protocol analysis breaks randomization in that characteristics of
study completers in intervention and control groups can differ, for
example, because participants in placebo groups may withdraw pri-
marily because of inefficacy and patients in drug groups may with-
draw more because of adverse effects.85
Effect sizes for relapse prevention were generally larger than for
acute therapy with the same drugs that are given to a mixture of re-
sponders and nonresponders, lending strong support for continu-
ation treatment, but long-term treatment might sensitize brain re-
ceptors, facilitating relapse after abrupt withdrawal.86Psychotherapy
offers a more optimistic perspective because it could cure by induc-
ing lasting cognitive and behavioral changes. Indeed, the few psy-
chotherapy meta-analyses that examined follow-up after the study
ended found that treatment effects persisted.
Evidence for publication bias was numerically more frequent
in pharmacotherapy meta-analyses, where it is a well-known
phenomenon,1 5although it also occurs in psychotherapy
research.16Because the main driver for publication bias is trial fail-
ure and blind trials are more likely to fail, the infrequent use of
blinding in psychotherapy trials is important. Finally, the mean
baseline severity in psychotherapy trials on MDD was lower than
that in antidepressant trials. This could mean that psychotherapy
is particularly efficacious in milder forms of MDD in which antide-
pressants are less effective.4
Strengths and Limitations
In our study, the criteria of a systematic review were met, but obvi-
ously there are multiple limitations. Because the selection of the
meta-analyses was most prone to bias, we described this process
in detail (Supplement [eAppendix 5]). We always compared phar-
macotherapy and psychotherapy for the same disorders, and we
strongly discourage comparisons of treatments across disorders. Still,
meta-analyses depend on the quality of the included studies. To be
systematic, we chose the most recent meta-analyses because older
ones, although sometimes methodologically better, would have been
out of date. We analyzed classes of drugs rather than single agents,
assuming that the original authors had made an appropriate deci-
sion to pooldrugs. Therefore, our results are not specific, al-
though, with few exceptions, no large efficacy differences exist within
drug classes. We preferred psychotherapies under the umbrellas of
CBT and psychodynamic therapy; occasionally, more efficacious psy-
chotherapies may have been missed. We also did not examine ef-
fective interventions such as psychoeducation.87,88
To examine ad-
verse effects and balance them with efficacy was impossible, but this
domain is a major advantage of psychotherapy. Although based on
RCTs, meta-analysis itself is an observational method with many limi-
tations (eg, “garbage in garbage out” and heterogeneity), and RCTs
lack generalizability. Finally, the interpretation of effect size is con-
troversial. Therefore, we emphasize the importance of reading the
actual numbers in the Supplement (eAppendix 6).89However, meta-
analysis served as a useful tool to systematically provide a rough over-
view of what benefit psychiatry can offer patients. Even if other au-
thors made different choices, the chance of them reaching essentially
different conclusions is unlikely.
Conclusions
Because of the multiple methodologic differences between phar-
macotherapy and psychotherapy trials, indirect comparisons are
problematic. Meta-analysts should consistently address quality
indicators, such as control groups or degree of blinding. Head-to-
head trials are scarce, and funding for them is urgently needed.
Research should focus on how pharmacotherapy and psycho-
therapy can best be combined to obtain maximally synergistic
effects rather than debate the use of one treatment over the
other.
Clinical Review & EducationReview Pharmacotherapy and Psychotherapy in Adults
712 JAMA PsychiatryJune 2014Volume 71, Number 6 jamapsychiatry.com
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

Copyright 2014 American Medical Association. All rights reserved.
ARTICLE INFORMATION
Submitted for Publication: July 1, 2013; final
revision received January 24, 2014; accepted
January 24, 2014.
Published Online: April 30, 2014.
doi:10.1001/jamapsychiatry.2014.112.
Author Contributions: Dr Huhn had full access to
all the data in the study and takes responsibility for
the integrity of the data and the accuracy of the
data analysis.
Study concept and design: Huhn, Förstl, Davis, S.
Leucht.
Acquisition, analysis, or interpretation of data: All
authors.
Drafting of the manuscript: Huhn, S. Leucht.
Critical revision of the manuscript for important
intellectual content: All authors.
Statistical analysis: Huhn, Spineli, Samara, Davis, S.
Leucht.
Obtained funding: S. Leucht.
Administrative, technical, or material support: C.
Leucht, Samara, Dold.
Study supervision: Kissling, Förstl, Davis, S. Leucht.
Conflict of Interest Disclosures: Dr Kissling has
received speaker and/or advisory board and
consultancy honoraria from Janssen,
sanofi-aventis, Johnson & Johnson, Pfizer, Bayer,
BMS, AstraZeneca, Lundbeck, Novartis, and Eli Lilly.
Dr Förstl has received speaker fees and honoraria
for serving on advisory boards from all major
international drug companies. Dr Pitschel-Walz has
received lecture honoraria from AstraZeneca, Eli
Lilly, Janssen, Pfizer, and sanofi-aventis as well as
honoraria for advisory boards from Janssen. Dr C.
Leucht is Dr. S. Leucht’s spouse and shares his
conflicts of interest. Dr S. Leucht has received
honoraria for lectures from Abbvie, AstraZeneca,
Bristol-Myers Squibb, ICON, Eli Lilly, Janssen,
Johnson & Johnson, Roche, sanofi-aventis,
Lundbeck, and Pfizer as well as honoraria for
serving on consulting/advisory boards from Roche,
Eli Lilly, Medavante, Bristol-Myers Squibb,
Alkermes, Janssen, Johnson & Johnson, and
Lundbeck. Eli Lilly provided medication for a trial in
which Dr Leucht was the primary investigator. No
other conflicts were disclosed.
Additional Contributions: Pim Cuijpers, PhD
(Department of Clinical Psychology, Vrije
Universiteit, Amsterdam, the Netherlands), Molly
Magill, PhD (Department of Behavioral and Social
Sciences, Brown University Medical School,
Providence, Rhode Island), Glen Spielmans, PhD
(Department of Psychology, Metropolitan State
University, St Paul, Minnesota), Ayseguel Yildiz, MD
(Department of Psychiatry, Dokuz Eylül University,
Izmir, Turkey), Erick Turner, MD (Departments of
Psychiatry and Pharmacology, Oregon Health and
Science University, Portland, Oregon), and Peter
McKenna, MD (FIDMAG Research Foundation,
Germanes Hospitalàries, Barcelona, Spain;
CIBERSAM, Spain) provided additional data.
Bartosz Helfer, MSc, Sarah Longhi, MD (both with
the Department of Psychiatry and Psychotherapy,
Technische Universität München, München,
Germany), and Sabrina Reed, medical student
(College of Medicine, University of Illinois at
Chicago), helped with statistical calculation and
data extraction. No contributor received financial
compensation.
REFERENCES
1.Morrison AP, Hutton P, Shiers D, Turkington D.
Antipsychotics: is it time to introduce patient
choice? Br J Psychiatry. 2012;201:83-84.
2. Kaduszkiewicz H, Zimmermann T,
Beck-Bornholdt HP, van den Bussche H.
Cholinesterase inhibitors for patients with
Alzheimer’s disease: systematic review of
randomised clinical trials. BMJ. 2005;331(7512):321-
327.
3. Moncrieff J. Lithium revisited: a re-examination
of the placebo-controlled trials of lithium
prophylaxis in manic-depressive disorder. Br J
Psychiatry. 1995;167(5):569-574.
4. Kirsch I, Deacon BJ, Huedo-Medina TB, Scoboria
A, Moore TJ, Johnson BT. Initial severity and
antidepressant benefits: a meta-analysis of data
submitted to the Food and Drug Administration.
PLoS Med. 2008;5(2):e45. doi:10.1371/journal
.pmed.0050045.
5. Leucht S, Hierl S, Kissling W, Dold M, Davis JM.
Putting the efficacy of psychiatric and general
medicine medication into perspective: review of
meta-analyses. Br J Psychiatry. 2012;200(2):97-
106.
6. Thombs BD, Jewett LR, Bassel M. Is there room
for criticism of studies of psychodynamic
psychotherapy? Am Psychol. 2011;66(2):148-149.
7. Greenberg G. Manufacturing Depression: The
Secret History of a Modern Disease. New York, NY:
Simon & Schuster; 2010.
8. Cohen J. Statistical Power Analysis for the
Behavioral Sciences. 2nd ed. Hillsdale, NJ: Lawrence
Erlbaum Associates; 1988.
9. Moher D, Liberati A, Tetzlaff J, Altman DG;
PRISMA Group. Preferred reporting items for
systematic reviews and meta-analyses: the PRISMA
statement. PLoS Med. 2009;6(7):e1000097.
doi:10.1371/journal.pmed.1000097.
10.National Institute for Health and Clinical
Excellence (NICE) guidelines. http://guidance.nice-
.org.uk/CG/Published. Accessed October 14, 2013.
11.Review Manager RevMan [computer program].
Version 5.0.22. Copenhagen, Denmark: Cochrane
Collaboration; 2008.
12.Comprehensive Meta-analysis [computer
program]. Version 2. Englewood, NJ: Biostat Inc;
2006.
13.Higgins JPT, Green S, eds. Cochrane Handbook
for Systematic Reviews of Interventions. Version
5.1.0. Copenhagen, Denmark: Cochrane
Collaboration; March 2011.
14.Shea BJ, Hamel C, Wells GA, et al. AMSTAR is a
reliable and valid measurement tool to assess the
methodological quality of systematic reviews. J Clin
Epidemiol. 2009;62(10):1013-1020.
15.Turner EH, Matthews AM, Linardatos E, Tell RA,
Rosenthal R. Selective publication of
antidepressant trials and its influence on apparent
efficacy. N Engl J Med. 2008;358(3):252-260.
16.Cuijpers P, Smit F, Bohlmeijer E, Hollon SD,
Andersson G. Efficacy of cognitive-behavioural
therapy and other psychological treatments for
adult depression: meta-analytic study of
publication bias. Br J Psychiatry. 2010;196(3):173-
178.
17.Bacaltchuk J, Hay P. Antidepressants versus
placebo for people with bulimia nervosa. Cochrane
Database Syst Rev. 2003;(4):CD003391. doi:10
.1002/14651858.CD003391.
18.Birks J. Cholinesterase inhibitors for
Alzheimer’s disease. Cochrane Database Syst Rev.
2006;(1):CD005593. doi:10.1002/14651858
.CD005593.
19.Bloch MH, Landeros-Weisenberger A,
Dombrowski P, et al. Systematic review:
pharmacological and behavioral treatment for
trichotillomania. Biol Psychiatry. 2007;62(8):839-
846.
20. Buscemi N, Vandermeer B, Friesen C, et al. The
efficacy and safety of drug treatments for chronic
insomnia in adults: a meta-analysis of RCTs. J Gen
Intern Med. 2007;22(9):1335-1350.
21.Castells X, Ramos-Quiroga JA, Bosch R,
Nogueira M, Casas M. Amphetamines for attention
deficit hyperactivity disorder (ADHD) in adults.
Cochrane Database Syst Rev. 2011;(6):CD007813.
doi:10.1002/14651858.CD007813.
22.Claudino AM, Hay P, Lima MS, Bacaltchuk J,
Schmidt U, Treasure J. Antidepressants for anorexia
nervosa. Cochrane Database Syst Rev. 2006;(1):
CD004365.
23.Donovan MR, Glue P, Kolluri S, Emir B.
Comparative efficacy of antidepressants in
preventing relapse in anxiety disorders—a
meta-analysis. J Affect Disord. 2010;123(1-3):9-16.
24. Fishbain DA, Cutler RB, Rosomoff HL,
Rosomoff RS. Do antidepressants have an analgesic
effect in psychogenic pain and somatoform pain
disorder? a meta-analysis. Psychosom Med. 1998;
60(4):503-509.
25.Glue P, Donovan MR, Kolluri S, Emir B.
Meta-analysis of relapse prevention antidepressant
trials in depressive disorders. Aust N Z J Psychiatry.
2010;44(8):697-705.
26. Holbrook AM, Crowther R, Lotter A, Cheng C,
King D. Meta-analysis of benzodiazepine use in the
treatment of insomnia. CMAJ. 2000;162(2):225-
233.
27.Ipser JC, Kariuki CM, Stein DJ.
Pharmacotherapy for social anxiety disorder:
a systematic review. Expert Rev Neurother. 2008;8
(2):235-257.
28. Kavirajan H, Schneider LS. Efficacy and adverse
effects of cholinesterase inhibitors and memantine
in vascular dementia: a meta-analysis of
randomised controlled trials. Lancet Neurol. 2007;6
(9):782-792.
29. Kishi T, Kafantaris V, Sunday S, Sheridan EM,
Correll CU. Are antipsychotics effective for the
treatment of anorexia nervosa? results from a
systematic review and meta-analysis. J Clin
Psychiatry. 2012;73(6):e757-e766. doi:10.4088
/JCP.12r07691.
30. Leucht S, Arbter D, Engel RR, Kissling W, Davis
JM. How effective are second-generation
antipsychotic drugs? a meta-analysis of
placebo-controlled trials. Mol Psychiatry. 2009;14
(4):429-447.
31.Leucht S, Tardy M, Komossa K, et al.
Antipsychotic drugs versus placebo for relapse
Pharmacotherapy and Psychotherapy in Adults Review Clinical Review & Education
jamapsychiatry.com JAMA PsychiatryJune 2014Volume 71, Number 6 713
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
ARTICLE INFORMATION
Submitted for Publication: July 1, 2013; final
revision received January 24, 2014; accepted
January 24, 2014.
Published Online: April 30, 2014.
doi:10.1001/jamapsychiatry.2014.112.
Author Contributions: Dr Huhn had full access to
all the data in the study and takes responsibility for
the integrity of the data and the accuracy of the
data analysis.
Study concept and design: Huhn, Förstl, Davis, S.
Leucht.
Acquisition, analysis, or interpretation of data: All
authors.
Drafting of the manuscript: Huhn, S. Leucht.
Critical revision of the manuscript for important
intellectual content: All authors.
Statistical analysis: Huhn, Spineli, Samara, Davis, S.
Leucht.
Obtained funding: S. Leucht.
Administrative, technical, or material support: C.
Leucht, Samara, Dold.
Study supervision: Kissling, Förstl, Davis, S. Leucht.
Conflict of Interest Disclosures: Dr Kissling has
received speaker and/or advisory board and
consultancy honoraria from Janssen,
sanofi-aventis, Johnson & Johnson, Pfizer, Bayer,
BMS, AstraZeneca, Lundbeck, Novartis, and Eli Lilly.
Dr Förstl has received speaker fees and honoraria
for serving on advisory boards from all major
international drug companies. Dr Pitschel-Walz has
received lecture honoraria from AstraZeneca, Eli
Lilly, Janssen, Pfizer, and sanofi-aventis as well as
honoraria for advisory boards from Janssen. Dr C.
Leucht is Dr. S. Leucht’s spouse and shares his
conflicts of interest. Dr S. Leucht has received
honoraria for lectures from Abbvie, AstraZeneca,
Bristol-Myers Squibb, ICON, Eli Lilly, Janssen,
Johnson & Johnson, Roche, sanofi-aventis,
Lundbeck, and Pfizer as well as honoraria for
serving on consulting/advisory boards from Roche,
Eli Lilly, Medavante, Bristol-Myers Squibb,
Alkermes, Janssen, Johnson & Johnson, and
Lundbeck. Eli Lilly provided medication for a trial in
which Dr Leucht was the primary investigator. No
other conflicts were disclosed.
Additional Contributions: Pim Cuijpers, PhD
(Department of Clinical Psychology, Vrije
Universiteit, Amsterdam, the Netherlands), Molly
Magill, PhD (Department of Behavioral and Social
Sciences, Brown University Medical School,
Providence, Rhode Island), Glen Spielmans, PhD
(Department of Psychology, Metropolitan State
University, St Paul, Minnesota), Ayseguel Yildiz, MD
(Department of Psychiatry, Dokuz Eylül University,
Izmir, Turkey), Erick Turner, MD (Departments of
Psychiatry and Pharmacology, Oregon Health and
Science University, Portland, Oregon), and Peter
McKenna, MD (FIDMAG Research Foundation,
Germanes Hospitalàries, Barcelona, Spain;
CIBERSAM, Spain) provided additional data.
Bartosz Helfer, MSc, Sarah Longhi, MD (both with
the Department of Psychiatry and Psychotherapy,
Technische Universität München, München,
Germany), and Sabrina Reed, medical student
(College of Medicine, University of Illinois at
Chicago), helped with statistical calculation and
data extraction. No contributor received financial
compensation.
REFERENCES
1.Morrison AP, Hutton P, Shiers D, Turkington D.
Antipsychotics: is it time to introduce patient
choice? Br J Psychiatry. 2012;201:83-84.
2. Kaduszkiewicz H, Zimmermann T,
Beck-Bornholdt HP, van den Bussche H.
Cholinesterase inhibitors for patients with
Alzheimer’s disease: systematic review of
randomised clinical trials. BMJ. 2005;331(7512):321-
327.
3. Moncrieff J. Lithium revisited: a re-examination
of the placebo-controlled trials of lithium
prophylaxis in manic-depressive disorder. Br J
Psychiatry. 1995;167(5):569-574.
4. Kirsch I, Deacon BJ, Huedo-Medina TB, Scoboria
A, Moore TJ, Johnson BT. Initial severity and
antidepressant benefits: a meta-analysis of data
submitted to the Food and Drug Administration.
PLoS Med. 2008;5(2):e45. doi:10.1371/journal
.pmed.0050045.
5. Leucht S, Hierl S, Kissling W, Dold M, Davis JM.
Putting the efficacy of psychiatric and general
medicine medication into perspective: review of
meta-analyses. Br J Psychiatry. 2012;200(2):97-
106.
6. Thombs BD, Jewett LR, Bassel M. Is there room
for criticism of studies of psychodynamic
psychotherapy? Am Psychol. 2011;66(2):148-149.
7. Greenberg G. Manufacturing Depression: The
Secret History of a Modern Disease. New York, NY:
Simon & Schuster; 2010.
8. Cohen J. Statistical Power Analysis for the
Behavioral Sciences. 2nd ed. Hillsdale, NJ: Lawrence
Erlbaum Associates; 1988.
9. Moher D, Liberati A, Tetzlaff J, Altman DG;
PRISMA Group. Preferred reporting items for
systematic reviews and meta-analyses: the PRISMA
statement. PLoS Med. 2009;6(7):e1000097.
doi:10.1371/journal.pmed.1000097.
10.National Institute for Health and Clinical
Excellence (NICE) guidelines. http://guidance.nice-
.org.uk/CG/Published. Accessed October 14, 2013.
11.Review Manager RevMan [computer program].
Version 5.0.22. Copenhagen, Denmark: Cochrane
Collaboration; 2008.
12.Comprehensive Meta-analysis [computer
program]. Version 2. Englewood, NJ: Biostat Inc;
2006.
13.Higgins JPT, Green S, eds. Cochrane Handbook
for Systematic Reviews of Interventions. Version
5.1.0. Copenhagen, Denmark: Cochrane
Collaboration; March 2011.
14.Shea BJ, Hamel C, Wells GA, et al. AMSTAR is a
reliable and valid measurement tool to assess the
methodological quality of systematic reviews. J Clin
Epidemiol. 2009;62(10):1013-1020.
15.Turner EH, Matthews AM, Linardatos E, Tell RA,
Rosenthal R. Selective publication of
antidepressant trials and its influence on apparent
efficacy. N Engl J Med. 2008;358(3):252-260.
16.Cuijpers P, Smit F, Bohlmeijer E, Hollon SD,
Andersson G. Efficacy of cognitive-behavioural
therapy and other psychological treatments for
adult depression: meta-analytic study of
publication bias. Br J Psychiatry. 2010;196(3):173-
178.
17.Bacaltchuk J, Hay P. Antidepressants versus
placebo for people with bulimia nervosa. Cochrane
Database Syst Rev. 2003;(4):CD003391. doi:10
.1002/14651858.CD003391.
18.Birks J. Cholinesterase inhibitors for
Alzheimer’s disease. Cochrane Database Syst Rev.
2006;(1):CD005593. doi:10.1002/14651858
.CD005593.
19.Bloch MH, Landeros-Weisenberger A,
Dombrowski P, et al. Systematic review:
pharmacological and behavioral treatment for
trichotillomania. Biol Psychiatry. 2007;62(8):839-
846.
20. Buscemi N, Vandermeer B, Friesen C, et al. The
efficacy and safety of drug treatments for chronic
insomnia in adults: a meta-analysis of RCTs. J Gen
Intern Med. 2007;22(9):1335-1350.
21.Castells X, Ramos-Quiroga JA, Bosch R,
Nogueira M, Casas M. Amphetamines for attention
deficit hyperactivity disorder (ADHD) in adults.
Cochrane Database Syst Rev. 2011;(6):CD007813.
doi:10.1002/14651858.CD007813.
22.Claudino AM, Hay P, Lima MS, Bacaltchuk J,
Schmidt U, Treasure J. Antidepressants for anorexia
nervosa. Cochrane Database Syst Rev. 2006;(1):
CD004365.
23.Donovan MR, Glue P, Kolluri S, Emir B.
Comparative efficacy of antidepressants in
preventing relapse in anxiety disorders—a
meta-analysis. J Affect Disord. 2010;123(1-3):9-16.
24. Fishbain DA, Cutler RB, Rosomoff HL,
Rosomoff RS. Do antidepressants have an analgesic
effect in psychogenic pain and somatoform pain
disorder? a meta-analysis. Psychosom Med. 1998;
60(4):503-509.
25.Glue P, Donovan MR, Kolluri S, Emir B.
Meta-analysis of relapse prevention antidepressant
trials in depressive disorders. Aust N Z J Psychiatry.
2010;44(8):697-705.
26. Holbrook AM, Crowther R, Lotter A, Cheng C,
King D. Meta-analysis of benzodiazepine use in the
treatment of insomnia. CMAJ. 2000;162(2):225-
233.
27.Ipser JC, Kariuki CM, Stein DJ.
Pharmacotherapy for social anxiety disorder:
a systematic review. Expert Rev Neurother. 2008;8
(2):235-257.
28. Kavirajan H, Schneider LS. Efficacy and adverse
effects of cholinesterase inhibitors and memantine
in vascular dementia: a meta-analysis of
randomised controlled trials. Lancet Neurol. 2007;6
(9):782-792.
29. Kishi T, Kafantaris V, Sunday S, Sheridan EM,
Correll CU. Are antipsychotics effective for the
treatment of anorexia nervosa? results from a
systematic review and meta-analysis. J Clin
Psychiatry. 2012;73(6):e757-e766. doi:10.4088
/JCP.12r07691.
30. Leucht S, Arbter D, Engel RR, Kissling W, Davis
JM. How effective are second-generation
antipsychotic drugs? a meta-analysis of
placebo-controlled trials. Mol Psychiatry. 2009;14
(4):429-447.
31.Leucht S, Tardy M, Komossa K, et al.
Antipsychotic drugs versus placebo for relapse
Pharmacotherapy and Psychotherapy in Adults Review Clinical Review & Education
jamapsychiatry.com JAMA PsychiatryJune 2014Volume 71, Number 6 713
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015

Copyright 2014 American Medical Association. All rights reserved.
prevention in schizophrenia: a systematic review
and meta-analysis. Lancet. 2012;379(9831):2063-
2071.
32.Levkovitz Y, Tedeschini E, Papakostas GI.
Efficacy of antidepressants for dysthymia:
a meta-analysis of placebo-controlled randomized
trials. J Clin Psychiatry. 2011;72(4):509-514.
33.Mattick RP, Breen C, Kimber J, Davoli M.
Methadone maintenance therapy vs no opioid
replacement therapy for opioid dependence.
Cochrane Database Syst Rev. 2009;(3):CD002209.
doi:10.1002/14651858.CD002209.
34. Mattick RP, Kimber J, Breen C, Davoli M.
Buprenorphine maintenance vs placebo or
methadone maintenance for opioid dependence.
Cochrane Database Syst Rev. 2008;(2):CD002207.
doi:10.1002/14651858.CD002207.
35.McShane R, Areosa Sastre A, Minakaran N.
Memantine for dementia. Cochrane Database Syst
Rev. 2006;(2):CD003154. doi:10.1002/14651858
.CD003154.
36. Melander H, Salmonson T, Abadie E, van
Zwieten-Boot B. A regulatory apologia—a review of
placebo-controlled studies in regulatory
submissions of new-generation antidepressants.
Eur Neuropsychopharmacol. 2008;18(9):623-627.
37.Mitte K. A meta-analysis of the efficacy of
psycho- and pharmacotherapy in panic disorder
with and without agoraphobia. J Affect Disord.
2005;88(1):27-45.
38. Mitte K, Noack P, Steil R, Hautzinger M. A
meta-analytic review of the efficacy of drug
treatment in generalized anxiety disorder. J Clin
Psychopharmacol. 2005;25(2):141-150.
39. Reas DL, Grilo CM. Review and meta-analysis of
pharmacotherapy for binge-eating disorder. Obesity
(Silver Spring). 2008;16(9):2024-2038.
40. Rösner S, Hackl-Herrwerth A, Leucht S, Lehert
P, Vecchi S, Soyka M. Acamprosate for alcohol
dependence. Cochrane Database Syst Rev. 2010;
(9):CD004332. doi:10.1002/14651858.CD004322.
41.Rösner S, Hackl-Herrwerth A, Leucht S, Vecchi
S, Srisurapanont M, Soyka M. Opioid antagonists for
alcohol dependence. Cochrane Database Syst Rev.
2010;(12):CD001867. doi:10.1002/14651858
.CD001867.
42. Sidor MM, Macqueen GM. Antidepressants for
the acute treatment of bipolar depression:
a systematic review and meta-analysis. J Clin
Psychiatry. 2011;72(2):156-167.
43. Soomro GM, Altman D, Rajagopal S,
Oakley-Browne M. Selective serotonin re-uptake
inhibitors (SSRIs) versus placebo for obsessive
compulsive disorder (OCD). Cochrane Database
Syst Rev. 2008;(1):CD001765. doi:10.1002
/14651858.CD001765.
44. Stein DJ, Ipser JC, Seedat S. Pharmacotherapy
for post traumatic stress disorder (PTSD). Cochrane
Database Syst Rev. 2006;(1):CD002795. doi:10
.1002/14651858.CD002795.
45. Van Lieshout RJ, MacQueen GM. Efficacy and
acceptability of mood stabilisers in the treatment of
acute bipolar depression: systematic review. Br J
Psychiatry. 2010;196(4):266-273.
46. Vieta E, Günther O, Locklear J, et al.
Effectiveness of psychotropic medications in the
maintenance phase of bipolar disorder:
a meta-analysis of randomized controlled trials. Int J
Neuropsychopharmacol. 2011;14(8):1029-1049.
47.Vita A, De Peri L, Sacchetti E. Antipsychotics,
antidepressants, anticonvulsants, and placebo on
the symptom dimensions of borderline personality
disorder: a meta-analysis of randomized controlled
and open-label trials. J Clin Psychopharmacol. 2011;
31(5):613-624.
48. Yildiz A, Vieta E, Leucht S, Baldessarini RJ.
Efficacy of antimanic treatments: meta-analysis of
randomized, controlled trials. Neuropsychopharma-
cology. 2011;36(2):375-389.
49. Cuijpers P, van Straten A, Schuurmans J, van
Oppen P, Hollon SD, Andersson G. Psychotherapy
for chronic major depression and dysthymia:
a meta-analysis. Clin Psychol Rev. 2010;30(1):51-62.
50. Driessen E, Cuijpers P, de Maat SCM, Abbass
AA, de Jonghe F, Dekker JJ. The efficacy of
short-term psychodynamic psychotherapy for
depression: a meta-analysis. Clin Psychol Rev. 2010;
30(1):25-36.
51.Dutra L, Stathopoulou G, Basden SL, Leyro TM,
Powers MB, Otto MW. A meta-analytic review of
psychosocial interventions for substance use
disorders. Am J Psychiatry. 2008;165(2):179-187.
52.Fisher CA, Hetrick SE, Rushford N. Family
therapy for anorexia nervosa. Cochrane Database
Syst Rev. 2010;(4):CD004780. doi:10.1002
/14651858.CD004780.
53.Hay PP, Bacaltchuk J, Stefano S, Kashyap P.
Psychological treatments for bulimia nervosa and
binging. Cochrane Database Syst Rev. 2009;(4):
CD000562. doi:10.1002/14651858.CD00562.
54. Hofmann SG, Smits JAJ. Cognitive-behavioral
therapy for adult anxiety disorders: a meta-analysis
of randomized placebo-controlled trials. J Clin
Psychiatry. 2008;69(4):621-632.
55.Irwin MR, Cole JC, Nicassio PM. Comparative
meta-analysis of behavioral interventions for
insomnia and their efficacy in middle-aged adults
and in older adults 55+ years of age. Health Psychol.
2006;25(1):3-14.
56. Kleinstäuber M, Witthöft M, Hiller W. Efficacy
of short-term psychotherapy for multiple medically
unexplained physical symptoms: a meta-analysis.
Clin Psychol Rev. 2011;31(1):146-160.
57.Lynch D, Laws KR, McKenna PJ. Cognitive
behavioural therapy for major psychiatric disorder:
does it really work? a meta-analytical review of
well-controlled trials. Psychol Med. 2010;40(1):9-
24.
58. Magill M, Ray LA. Cognitive-behavioral
treatment with adult alcohol and illicit drug users:
a meta-analysis of randomized controlled trials.
J Stud Alcohol Drugs. 2009;70(4):516-527.
59. Malmberg L, Fenton M. Individual
psychodynamic psychotherapy and psychoanalysis
for schizophrenia and severe mental illness.
Cochrane Database Syst Rev. 2001;(3):CD001360.
doi:10.1002/14651858.CD001360.
60. Olazarán J, Reisberg B, Clare L, et al.
Nonpharmacological therapies in Alzheimer’s
disease: a systematic review of efficacy. Dement
Geriatr Cogn Disord. 2010;30(2):161-178.
61.Stoffers JM, Völlm BA, Rücker G, Timmer A,
Huband N, Lieb K. Psychological therapies for
people with borderline personality disorder.
Cochrane Database Syst Rev. 2012;(8):CD005652.
doi:10.1002/14651858.CD005652.
62. Vasilaki EI, Hosier SG, Cox WM. The efficacy of
motivational interviewing as a brief intervention for
excessive drinking: a meta-analytic review. Alcohol
Alcohol. 2006;41(3):328-335.
63. Wolitzky-Taylor KB, Horowitz JD, Powers MB,
Telch MJ. Psychological approaches in the
treatment of specific phobias: a meta-analysis. Clin
Psychol Rev. 2008;28(6):1021-1037.
64. de Maat S, Dekker J, Schoevers R, de Jonghe F.
Relative efficacy of psychotherapy and
pharmacotherapy in the treatment of depression:
a meta-analysis. Psychother Res. 2006;16(5):566-
578.
65. Hay Phillipa PJ, Claudino Angélica M, Kaio
Marcel H. Antidepressants vs psychological
treatments and their combination for bulimia
nervosa. Cochrane Database Syst Rev. 2001;(4):
CD003385. doi:10.1002/14651858.CD003385.
66. Mitte K. Meta-analysis of cognitive-behavioral
treatments for generalized anxiety disorder:
a comparison with pharmacotherapy. Psychol Bull.
2005;131(5):785-795.
67. Spielmans GI, Berman MI, Usitalo AN.
Psychotherapy versus second-generation
antidepressants in the treatment of depression:
a meta-analysis. J Nerv Ment Dis. 2011;199(3):142-
149.
68. Bandelow B, Seidler-Brandler U, Becker A,
Wedekind D, Rüther E. Meta-analysis of
randomized controlled comparisons of
psychopharmacological and psychological
treatments for anxiety disorders. World J Biol
Psychiatry. 2007;8(3):175-187.
69. Amato L, Minozzi S, Davoli M, Vecchi S.
Psychosocial combined with agonist maintenance
treatments versus agonist maintenance treatments
alone for treatment of opioid dependence.
Cochrane Database Syst Rev. 2011;(10):CD004147.
doi:10.1002/14651858.CD004147.
70. Cuijpers P, Dekker J, Hollon SD, Andersson G.
Adding psychotherapy to pharmacotherapy in the
treatment of depressive disorders in adults:
a meta-analysis. J Clin Psychiatry. 2009;70(9):1219-
1229.
71.de Maat SM, Dekker J, Schoevers RA, de Jonghe
F. Relative efficacy of psychotherapy and combined
therapy in the treatment of depression:
a meta-analysis. Eur Psychiatry. 2007;22(1):1-8.
72.Furukawa TA, Watanabe N, Churchill R.
Combined psychotherapy plus antidepressants for
panic disorder with or without agoraphobia.
Cochrane Database Syst Rev. 2007;(1):CD004364.
doi:10.1002/14651858.CD004364.
73.Hetrick SE, Purcell R, Garner B, Parslow R.
Combined pharmacotherapy and psychological
therapies for post traumatic stress disorder (PTSD).
Cochrane Database Syst Rev. 2010;7(7):CD007316.
doi:10.1002/14651858.CD007316.
74.Jauhar S, McKenna PJ, Radua J, Fung E,
Salvador R, Laws KR. Cognitive-behavioural therapy
for the symptoms of schizophrenia: systematic
review and meta-analysis with examination of
potential bias. Br J Psychiatry. 2014;204(1):20-29.
75.Watanabe N, Churchill R, Furukawa TA.
Combined psychotherapy plus benzodiazepines for
panic disorder. Cochrane Database Syst Rev. 2009;
(1):CD005335. doi:10.1002/14651858.CD005335.
Clinical Review & EducationReview Pharmacotherapy and Psychotherapy in Adults
714 JAMA PsychiatryJune 2014Volume 71, Number 6 jamapsychiatry.com
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
prevention in schizophrenia: a systematic review
and meta-analysis. Lancet. 2012;379(9831):2063-
2071.
32.Levkovitz Y, Tedeschini E, Papakostas GI.
Efficacy of antidepressants for dysthymia:
a meta-analysis of placebo-controlled randomized
trials. J Clin Psychiatry. 2011;72(4):509-514.
33.Mattick RP, Breen C, Kimber J, Davoli M.
Methadone maintenance therapy vs no opioid
replacement therapy for opioid dependence.
Cochrane Database Syst Rev. 2009;(3):CD002209.
doi:10.1002/14651858.CD002209.
34. Mattick RP, Kimber J, Breen C, Davoli M.
Buprenorphine maintenance vs placebo or
methadone maintenance for opioid dependence.
Cochrane Database Syst Rev. 2008;(2):CD002207.
doi:10.1002/14651858.CD002207.
35.McShane R, Areosa Sastre A, Minakaran N.
Memantine for dementia. Cochrane Database Syst
Rev. 2006;(2):CD003154. doi:10.1002/14651858
.CD003154.
36. Melander H, Salmonson T, Abadie E, van
Zwieten-Boot B. A regulatory apologia—a review of
placebo-controlled studies in regulatory
submissions of new-generation antidepressants.
Eur Neuropsychopharmacol. 2008;18(9):623-627.
37.Mitte K. A meta-analysis of the efficacy of
psycho- and pharmacotherapy in panic disorder
with and without agoraphobia. J Affect Disord.
2005;88(1):27-45.
38. Mitte K, Noack P, Steil R, Hautzinger M. A
meta-analytic review of the efficacy of drug
treatment in generalized anxiety disorder. J Clin
Psychopharmacol. 2005;25(2):141-150.
39. Reas DL, Grilo CM. Review and meta-analysis of
pharmacotherapy for binge-eating disorder. Obesity
(Silver Spring). 2008;16(9):2024-2038.
40. Rösner S, Hackl-Herrwerth A, Leucht S, Lehert
P, Vecchi S, Soyka M. Acamprosate for alcohol
dependence. Cochrane Database Syst Rev. 2010;
(9):CD004332. doi:10.1002/14651858.CD004322.
41.Rösner S, Hackl-Herrwerth A, Leucht S, Vecchi
S, Srisurapanont M, Soyka M. Opioid antagonists for
alcohol dependence. Cochrane Database Syst Rev.
2010;(12):CD001867. doi:10.1002/14651858
.CD001867.
42. Sidor MM, Macqueen GM. Antidepressants for
the acute treatment of bipolar depression:
a systematic review and meta-analysis. J Clin
Psychiatry. 2011;72(2):156-167.
43. Soomro GM, Altman D, Rajagopal S,
Oakley-Browne M. Selective serotonin re-uptake
inhibitors (SSRIs) versus placebo for obsessive
compulsive disorder (OCD). Cochrane Database
Syst Rev. 2008;(1):CD001765. doi:10.1002
/14651858.CD001765.
44. Stein DJ, Ipser JC, Seedat S. Pharmacotherapy
for post traumatic stress disorder (PTSD). Cochrane
Database Syst Rev. 2006;(1):CD002795. doi:10
.1002/14651858.CD002795.
45. Van Lieshout RJ, MacQueen GM. Efficacy and
acceptability of mood stabilisers in the treatment of
acute bipolar depression: systematic review. Br J
Psychiatry. 2010;196(4):266-273.
46. Vieta E, Günther O, Locklear J, et al.
Effectiveness of psychotropic medications in the
maintenance phase of bipolar disorder:
a meta-analysis of randomized controlled trials. Int J
Neuropsychopharmacol. 2011;14(8):1029-1049.
47.Vita A, De Peri L, Sacchetti E. Antipsychotics,
antidepressants, anticonvulsants, and placebo on
the symptom dimensions of borderline personality
disorder: a meta-analysis of randomized controlled
and open-label trials. J Clin Psychopharmacol. 2011;
31(5):613-624.
48. Yildiz A, Vieta E, Leucht S, Baldessarini RJ.
Efficacy of antimanic treatments: meta-analysis of
randomized, controlled trials. Neuropsychopharma-
cology. 2011;36(2):375-389.
49. Cuijpers P, van Straten A, Schuurmans J, van
Oppen P, Hollon SD, Andersson G. Psychotherapy
for chronic major depression and dysthymia:
a meta-analysis. Clin Psychol Rev. 2010;30(1):51-62.
50. Driessen E, Cuijpers P, de Maat SCM, Abbass
AA, de Jonghe F, Dekker JJ. The efficacy of
short-term psychodynamic psychotherapy for
depression: a meta-analysis. Clin Psychol Rev. 2010;
30(1):25-36.
51.Dutra L, Stathopoulou G, Basden SL, Leyro TM,
Powers MB, Otto MW. A meta-analytic review of
psychosocial interventions for substance use
disorders. Am J Psychiatry. 2008;165(2):179-187.
52.Fisher CA, Hetrick SE, Rushford N. Family
therapy for anorexia nervosa. Cochrane Database
Syst Rev. 2010;(4):CD004780. doi:10.1002
/14651858.CD004780.
53.Hay PP, Bacaltchuk J, Stefano S, Kashyap P.
Psychological treatments for bulimia nervosa and
binging. Cochrane Database Syst Rev. 2009;(4):
CD000562. doi:10.1002/14651858.CD00562.
54. Hofmann SG, Smits JAJ. Cognitive-behavioral
therapy for adult anxiety disorders: a meta-analysis
of randomized placebo-controlled trials. J Clin
Psychiatry. 2008;69(4):621-632.
55.Irwin MR, Cole JC, Nicassio PM. Comparative
meta-analysis of behavioral interventions for
insomnia and their efficacy in middle-aged adults
and in older adults 55+ years of age. Health Psychol.
2006;25(1):3-14.
56. Kleinstäuber M, Witthöft M, Hiller W. Efficacy
of short-term psychotherapy for multiple medically
unexplained physical symptoms: a meta-analysis.
Clin Psychol Rev. 2011;31(1):146-160.
57.Lynch D, Laws KR, McKenna PJ. Cognitive
behavioural therapy for major psychiatric disorder:
does it really work? a meta-analytical review of
well-controlled trials. Psychol Med. 2010;40(1):9-
24.
58. Magill M, Ray LA. Cognitive-behavioral
treatment with adult alcohol and illicit drug users:
a meta-analysis of randomized controlled trials.
J Stud Alcohol Drugs. 2009;70(4):516-527.
59. Malmberg L, Fenton M. Individual
psychodynamic psychotherapy and psychoanalysis
for schizophrenia and severe mental illness.
Cochrane Database Syst Rev. 2001;(3):CD001360.
doi:10.1002/14651858.CD001360.
60. Olazarán J, Reisberg B, Clare L, et al.
Nonpharmacological therapies in Alzheimer’s
disease: a systematic review of efficacy. Dement
Geriatr Cogn Disord. 2010;30(2):161-178.
61.Stoffers JM, Völlm BA, Rücker G, Timmer A,
Huband N, Lieb K. Psychological therapies for
people with borderline personality disorder.
Cochrane Database Syst Rev. 2012;(8):CD005652.
doi:10.1002/14651858.CD005652.
62. Vasilaki EI, Hosier SG, Cox WM. The efficacy of
motivational interviewing as a brief intervention for
excessive drinking: a meta-analytic review. Alcohol
Alcohol. 2006;41(3):328-335.
63. Wolitzky-Taylor KB, Horowitz JD, Powers MB,
Telch MJ. Psychological approaches in the
treatment of specific phobias: a meta-analysis. Clin
Psychol Rev. 2008;28(6):1021-1037.
64. de Maat S, Dekker J, Schoevers R, de Jonghe F.
Relative efficacy of psychotherapy and
pharmacotherapy in the treatment of depression:
a meta-analysis. Psychother Res. 2006;16(5):566-
578.
65. Hay Phillipa PJ, Claudino Angélica M, Kaio
Marcel H. Antidepressants vs psychological
treatments and their combination for bulimia
nervosa. Cochrane Database Syst Rev. 2001;(4):
CD003385. doi:10.1002/14651858.CD003385.
66. Mitte K. Meta-analysis of cognitive-behavioral
treatments for generalized anxiety disorder:
a comparison with pharmacotherapy. Psychol Bull.
2005;131(5):785-795.
67. Spielmans GI, Berman MI, Usitalo AN.
Psychotherapy versus second-generation
antidepressants in the treatment of depression:
a meta-analysis. J Nerv Ment Dis. 2011;199(3):142-
149.
68. Bandelow B, Seidler-Brandler U, Becker A,
Wedekind D, Rüther E. Meta-analysis of
randomized controlled comparisons of
psychopharmacological and psychological
treatments for anxiety disorders. World J Biol
Psychiatry. 2007;8(3):175-187.
69. Amato L, Minozzi S, Davoli M, Vecchi S.
Psychosocial combined with agonist maintenance
treatments versus agonist maintenance treatments
alone for treatment of opioid dependence.
Cochrane Database Syst Rev. 2011;(10):CD004147.
doi:10.1002/14651858.CD004147.
70. Cuijpers P, Dekker J, Hollon SD, Andersson G.
Adding psychotherapy to pharmacotherapy in the
treatment of depressive disorders in adults:
a meta-analysis. J Clin Psychiatry. 2009;70(9):1219-
1229.
71.de Maat SM, Dekker J, Schoevers RA, de Jonghe
F. Relative efficacy of psychotherapy and combined
therapy in the treatment of depression:
a meta-analysis. Eur Psychiatry. 2007;22(1):1-8.
72.Furukawa TA, Watanabe N, Churchill R.
Combined psychotherapy plus antidepressants for
panic disorder with or without agoraphobia.
Cochrane Database Syst Rev. 2007;(1):CD004364.
doi:10.1002/14651858.CD004364.
73.Hetrick SE, Purcell R, Garner B, Parslow R.
Combined pharmacotherapy and psychological
therapies for post traumatic stress disorder (PTSD).
Cochrane Database Syst Rev. 2010;7(7):CD007316.
doi:10.1002/14651858.CD007316.
74.Jauhar S, McKenna PJ, Radua J, Fung E,
Salvador R, Laws KR. Cognitive-behavioural therapy
for the symptoms of schizophrenia: systematic
review and meta-analysis with examination of
potential bias. Br J Psychiatry. 2014;204(1):20-29.
75.Watanabe N, Churchill R, Furukawa TA.
Combined psychotherapy plus benzodiazepines for
panic disorder. Cochrane Database Syst Rev. 2009;
(1):CD005335. doi:10.1002/14651858.CD005335.
Clinical Review & EducationReview Pharmacotherapy and Psychotherapy in Adults
714 JAMA PsychiatryJune 2014Volume 71, Number 6 jamapsychiatry.com
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide

Copyright 2014 American Medical Association. All rights reserved.
76. Acarturk C, Cuijpers P, van Straten A, de Graaf
R. Psychological treatment of social anxiety
disorder: a meta-analysis. Psychol Med. 2009;39
(2):241-254.
77.Bradley R, Greene J, Russ E, Dutra L, Westen D.
A multidimensional meta-analysis of psychotherapy
for PTSD. Am J Psychiatry. 2005;162(2):214-227.
78. Cuijpers P, van Straten A, Bohlmeijer E, Hollon
SD, Andersson G. The effects of psychotherapy for
adult depression are overestimated:
a meta-analysis of study quality and effect size.
Psychol Med. 2010;40(2):211-223.
79. Cuijpers P, van Straten A, Warmerdam L,
Andersson G. Psychotherapy versus the
combination of psychotherapy and
pharmacotherapy in the treatment of depression:
a meta-analysis. Depress Anxiety. 2009;26(3):279-
288.
80. Barbui C, Furukawa TA, Cipriani A.
Effectiveness of paroxetine in the treatment of
acute major depression in adults: a systematic
re-examination of published and unpublished data
from randomized trials. CMAJ. 2008;178(3):296-
305.
81.Trikalinos TA, Churchill R, Ferri M, et al; EU-PSI
project. Effect sizes in cumulative meta-analyses of
mental health randomized trials evolved over time.
J Clin Epidemiol. 2004;57(11):1124-1130.
82. Egger M, Davey Smith G, Schneider M, Minder
C. Bias in meta-analysis detected by a simple,
graphical test. BMJ. 1997;315(7109):629-634.
83. Tolin DF. Is cognitive-behavioral therapy more
effective than other therapies? a meta-analytic
review. Clin Psychol Rev. 2010;30(6):710-720.
84. Baethge C, Assall OP, Baldessarini RJ.
Systematic review of blinding assessment in
randomized controlled trials in schizophrenia and
affective disorders, 2000-2010. Psychother
Psychosom. 2013;82(3):152-160.
85. Mallinckrodt CH, Clark SW, Carroll RJ,
Molenbergh G. Assessing response profiles from
incomplete longitudinal clinical trial data under
regulatory considerations. J Biopharm Stat. 2003;13
(2):179-190.
86. Moncrieff J. Does antipsychotic withdrawal
provoke psychosis? review of the literature on rapid
onset psychosis (supersensitivity psychosis) and
withdrawal-related relapse. Acta Psychiatr Scand.
2006;114(1):3-13.
87.Colom F, Vieta E, Martinez-Aran A, et al.
A randomized trial on the efficacy of group
psychoeducation in the prophylaxis of recurrences
in bipolar patients whose disease is in remission.
Arch Gen Psychiatry. 2003;60(4):402-407.
88. Xia J, Merinder LB, Belgamwar MR.
Psychoeducation for schizophrenia. Cochrane
Database Syst Rev. 2011;(6):CD002831. doi:10
.1002/14651858.CD002831.
89. Concato J, Shah N, Horwitz RI. Randomized,
controlled trials, observational studies, and the
hierarchy of research designs. N Engl J Med. 2000;
342(25):1887-1892.
Pharmacotherapy and Psychotherapy in Adults Review Clinical Review & Education
jamapsychiatry.com JAMA PsychiatryJune 2014Volume 71, Number 6 715
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
76. Acarturk C, Cuijpers P, van Straten A, de Graaf
R. Psychological treatment of social anxiety
disorder: a meta-analysis. Psychol Med. 2009;39
(2):241-254.
77.Bradley R, Greene J, Russ E, Dutra L, Westen D.
A multidimensional meta-analysis of psychotherapy
for PTSD. Am J Psychiatry. 2005;162(2):214-227.
78. Cuijpers P, van Straten A, Bohlmeijer E, Hollon
SD, Andersson G. The effects of psychotherapy for
adult depression are overestimated:
a meta-analysis of study quality and effect size.
Psychol Med. 2010;40(2):211-223.
79. Cuijpers P, van Straten A, Warmerdam L,
Andersson G. Psychotherapy versus the
combination of psychotherapy and
pharmacotherapy in the treatment of depression:
a meta-analysis. Depress Anxiety. 2009;26(3):279-
288.
80. Barbui C, Furukawa TA, Cipriani A.
Effectiveness of paroxetine in the treatment of
acute major depression in adults: a systematic
re-examination of published and unpublished data
from randomized trials. CMAJ. 2008;178(3):296-
305.
81.Trikalinos TA, Churchill R, Ferri M, et al; EU-PSI
project. Effect sizes in cumulative meta-analyses of
mental health randomized trials evolved over time.
J Clin Epidemiol. 2004;57(11):1124-1130.
82. Egger M, Davey Smith G, Schneider M, Minder
C. Bias in meta-analysis detected by a simple,
graphical test. BMJ. 1997;315(7109):629-634.
83. Tolin DF. Is cognitive-behavioral therapy more
effective than other therapies? a meta-analytic
review. Clin Psychol Rev. 2010;30(6):710-720.
84. Baethge C, Assall OP, Baldessarini RJ.
Systematic review of blinding assessment in
randomized controlled trials in schizophrenia and
affective disorders, 2000-2010. Psychother
Psychosom. 2013;82(3):152-160.
85. Mallinckrodt CH, Clark SW, Carroll RJ,
Molenbergh G. Assessing response profiles from
incomplete longitudinal clinical trial data under
regulatory considerations. J Biopharm Stat. 2003;13
(2):179-190.
86. Moncrieff J. Does antipsychotic withdrawal
provoke psychosis? review of the literature on rapid
onset psychosis (supersensitivity psychosis) and
withdrawal-related relapse. Acta Psychiatr Scand.
2006;114(1):3-13.
87.Colom F, Vieta E, Martinez-Aran A, et al.
A randomized trial on the efficacy of group
psychoeducation in the prophylaxis of recurrences
in bipolar patients whose disease is in remission.
Arch Gen Psychiatry. 2003;60(4):402-407.
88. Xia J, Merinder LB, Belgamwar MR.
Psychoeducation for schizophrenia. Cochrane
Database Syst Rev. 2011;(6):CD002831. doi:10
.1002/14651858.CD002831.
89. Concato J, Shah N, Horwitz RI. Randomized,
controlled trials, observational studies, and the
hierarchy of research designs. N Engl J Med. 2000;
342(25):1887-1892.
Pharmacotherapy and Psychotherapy in Adults Review Clinical Review & Education
jamapsychiatry.com JAMA PsychiatryJune 2014Volume 71, Number 6 715
Copyright 2014 American Medical Association. All rights reserved.
Downloaded From: http://archpsyc.jamanetwork.com/ by a Ohio State University User on 02/08/2015
1 out of 10
Your All-in-One AI-Powered Toolkit for Academic Success.
+13062052269
info@desklib.com
Available 24*7 on WhatsApp / Email
![[object Object]](/_next/static/media/star-bottom.7253800d.svg)
Unlock your academic potential
Copyright © 2020–2026 A2Z Services. All Rights Reserved. Developed and managed by ZUCOL.