Comprehensive Cash Flow Analysis for Business Expansion Projects
VerifiedAdded on 2021/03/10
|6
|2484
|124
Homework Assignment
AI Summary
The assignment analyzes incremental cash flow, detailing initial investments, depreciation methods, and salvage value calculations. It explores scenarios like expanding product lines and launching new products, emphasizing the importance of incremental cash flow. The document defines key financial metrics such as the payback method, net present value (NPV), internal rate of return (IRR), and profitability index, explaining their roles in project evaluation. It also delves into concepts of market and corporate risk, and comparative advantage. Furthermore, the assignment covers comparative experiments in research and data collection methods, including the design of questionnaires. The document also summarizes key points of methods to follow determining the caloric content listed in nutritional labels of packaged food items under FDA regulations. Finally, the assignment includes references to relevant sources.
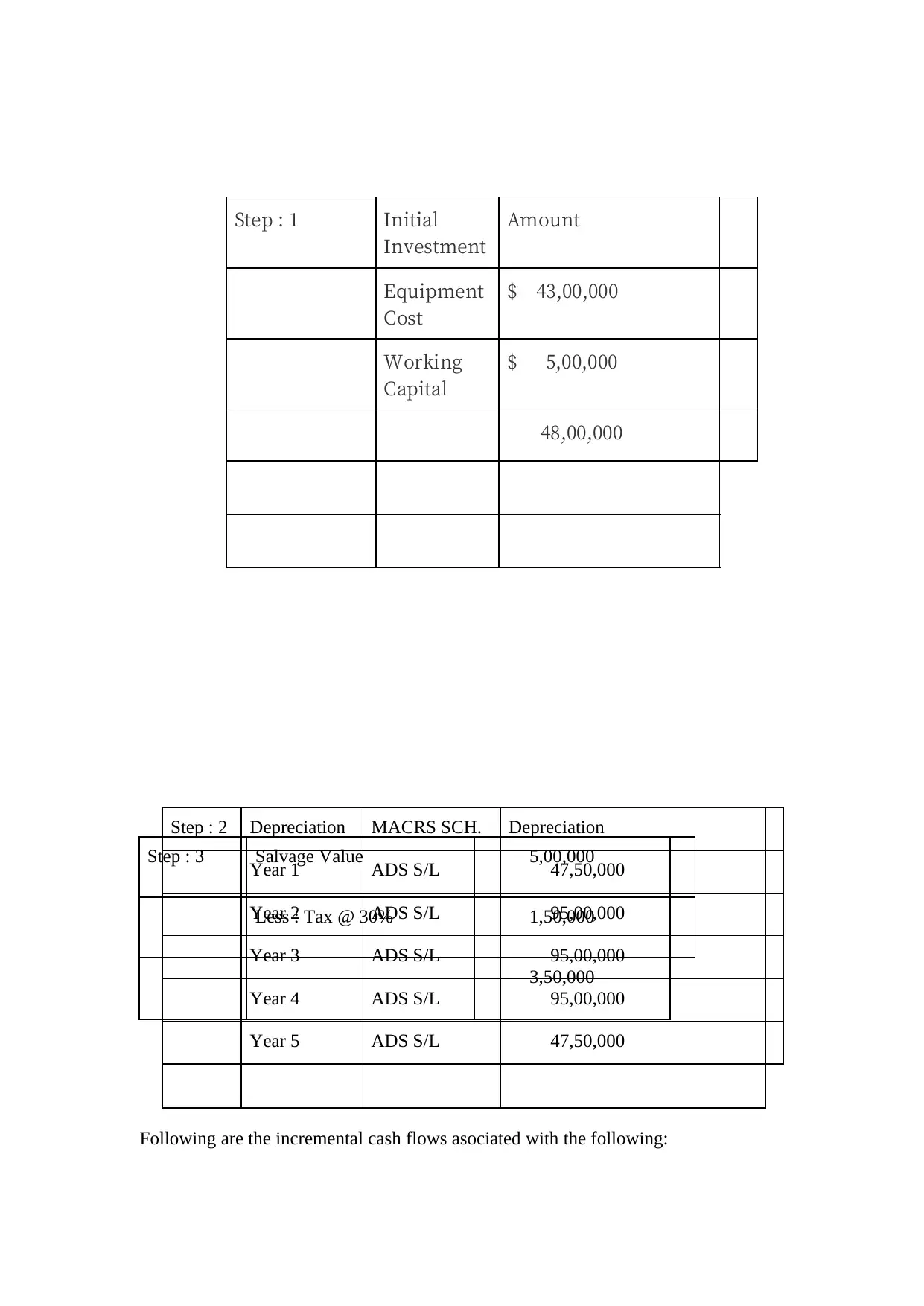
Step : 3 Salvage Value 5,00,000
Less : Tax @ 30% 1,50,000
3,50,000
Following are the incremental cash flows asociated with the following:
Step : 1 Initial
Investment
Amount
Equipment
Cost
$ 43,00,000
Working
Capital
$ 5,00,000
48,00,000
Step : 2 Depreciation MACRS SCH. Depreciation
Year 1 ADS S/L 47,50,000
Year 2 ADS S/L 95,00,000
Year 3 ADS S/L 95,00,000
Year 4 ADS S/L 95,00,000
Year 5 ADS S/L 47,50,000
Less : Tax @ 30% 1,50,000
3,50,000
Following are the incremental cash flows asociated with the following:
Step : 1 Initial
Investment
Amount
Equipment
Cost
$ 43,00,000
Working
Capital
$ 5,00,000
48,00,000
Step : 2 Depreciation MACRS SCH. Depreciation
Year 1 ADS S/L 47,50,000
Year 2 ADS S/L 95,00,000
Year 3 ADS S/L 95,00,000
Year 4 ADS S/L 95,00,000
Year 5 ADS S/L 47,50,000
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

a) Expanding a product line- In such a case we will have an initial investment which
is needed for expansion and is an outflow. The line would have its own revenues and
would require some expenditure as well for maintenance. So, incremental cash flow in
this case would be excess of revenue over expenditure and initial expansion cost.
b) Launching new product/service- For this purpose, company has to do some
expenditure on research and development to identify the financial prospects of
launching a new product. Accordingly such expenditure is an initial cash outlay for
the company. In case product is launched, it will have its own revenue and costs.
Thus, in this case incremental cash flows would be excess of product revenues over
product costs and initial investment.
1) An incremental cash flow refers to additional cash flow that the company makes by
undertaking a new project that could be expanding a product line or launching a new
product or service. An positive incremental cash flow means company's cash flow
will increase with acceptance of the project. There would be incremental cash flow for
the company by way of increase in sales by expanding a product line or launching a
new product or service.
2) Payback Method : Payback method is use to determine the period of time taken to
recover the cost of investment. For example, If Mr A investments in a project which
cost $500000 and the cashflow from project is $100000, the payback period would be
5 yrs since it would take 5yrs to recover $500000. ($500000/$100000).
Net Present Value : Net Present Value method refers to difference between the
present value of cash inflow and present value of cash outflow over a period of time.
The method is used to analyse the profitability or to make decision whether the
project should be undertaken or whether it is profitable. If net present value is positive
the project will be profitable for the company and negative net present value is not a
profitable project.
Internal Rate of Return : Internal rate of return is the discount rate that makes the
net present value of all cash flows from a project equal to zero. It is used to determine
the profitability of the project. If the internal rate of return exceeds the company's
required rate of return than the project is acceptable, if internal rate of return is less
than the company's required rate of return that the project is not acceptable.
Profitability Index Method : Profitability Index is an index which is determined by
dividing projects Present value of cash inflow by Initial investment. An index of less
than 1 refers to the present value of cash inflow is less than the cost of investment. An
index of more than 1 would be profitable for the project.
Present value of costs = $1500000
Net incremental annual benefits = $180000-$75000 = $105000
PV of total benefits =105000(P/A, 6%,25) +35000(P/F,6%,25) = $1,342,252.40+
$8,154.95= $1350407.35
is needed for expansion and is an outflow. The line would have its own revenues and
would require some expenditure as well for maintenance. So, incremental cash flow in
this case would be excess of revenue over expenditure and initial expansion cost.
b) Launching new product/service- For this purpose, company has to do some
expenditure on research and development to identify the financial prospects of
launching a new product. Accordingly such expenditure is an initial cash outlay for
the company. In case product is launched, it will have its own revenue and costs.
Thus, in this case incremental cash flows would be excess of product revenues over
product costs and initial investment.
1) An incremental cash flow refers to additional cash flow that the company makes by
undertaking a new project that could be expanding a product line or launching a new
product or service. An positive incremental cash flow means company's cash flow
will increase with acceptance of the project. There would be incremental cash flow for
the company by way of increase in sales by expanding a product line or launching a
new product or service.
2) Payback Method : Payback method is use to determine the period of time taken to
recover the cost of investment. For example, If Mr A investments in a project which
cost $500000 and the cashflow from project is $100000, the payback period would be
5 yrs since it would take 5yrs to recover $500000. ($500000/$100000).
Net Present Value : Net Present Value method refers to difference between the
present value of cash inflow and present value of cash outflow over a period of time.
The method is used to analyse the profitability or to make decision whether the
project should be undertaken or whether it is profitable. If net present value is positive
the project will be profitable for the company and negative net present value is not a
profitable project.
Internal Rate of Return : Internal rate of return is the discount rate that makes the
net present value of all cash flows from a project equal to zero. It is used to determine
the profitability of the project. If the internal rate of return exceeds the company's
required rate of return than the project is acceptable, if internal rate of return is less
than the company's required rate of return that the project is not acceptable.
Profitability Index Method : Profitability Index is an index which is determined by
dividing projects Present value of cash inflow by Initial investment. An index of less
than 1 refers to the present value of cash inflow is less than the cost of investment. An
index of more than 1 would be profitable for the project.
Present value of costs = $1500000
Net incremental annual benefits = $180000-$75000 = $105000
PV of total benefits =105000(P/A, 6%,25) +35000(P/F,6%,25) = $1,342,252.40+
$8,154.95= $1350407.35

Benefit Cost Ratio = Present value of Benefits/Present value of costs =
1350407.35/1500000 = 0.9003
Therefore, the benefit cost ratio for this capacity expansion is 0.9003
(B)
Option A is correct. Market Risk is measured by the project's beta. A project with a
higher beta bears more market risk. Here, project A has higher beta (1.20) than project
B and hence, it has higher market risk.
Option B is not correct. Stand-alone risk is measured by the standard deviation. Here,
project B has higher standard deviation ($130,000) and therefore has a higher stand-
alone risk as compared to A.
Option C is not correct. Corporate risk is measured by the correlation of the project's
cash flows relative to the firm's existing projects. Higher, the correlation, higher the
corporate risk. Since, project B has higher correlation (.80), it has more corporate risk
as compared to project A.
Comparative advantage is an economic law referring to the ability of an individual or
group to carry out a specific economic activity at a lower relative opportunity cost or
autarky price in comparison to other economic actors. In order to determine if
comparative advantages exist between the two workers, we have to figure out the
opportunity cost of making one unit of one of the items. The worker who has the
lowest opportunity costs of producing one unit of product has the comparative
advantage and thus must specialize in performing the task.
Comparative Experiments is testing two variables by designing a experiment.This can
be designed to collect data on different/objects/events or collect data under different
condition to test the hypothesis.
It contains a testable hypothesis where we have at least one independent variable and
one dependent variables and a control group. These experiments are usually designed
to test the relationships between independent and dependent variable. Data can be
collected here by both quantitatively and/or qualitatively to draw conclusions and can
support/reject the hypothesis.
Once the results are received of the comparative experiment, the scientist can
compare the difference in the dependent variable C for each (independent variable)
treatment, concluding either one (independent variable) treatment is more effective
than the other or that both independent variable(treatments) have the same
effectiveness.
(c)
Population - The director of market research wants to conduct the study of the time
spent on shopping for clothing by the working women of the area, working women of
the area are the population of interest.
1350407.35/1500000 = 0.9003
Therefore, the benefit cost ratio for this capacity expansion is 0.9003
(B)
Option A is correct. Market Risk is measured by the project's beta. A project with a
higher beta bears more market risk. Here, project A has higher beta (1.20) than project
B and hence, it has higher market risk.
Option B is not correct. Stand-alone risk is measured by the standard deviation. Here,
project B has higher standard deviation ($130,000) and therefore has a higher stand-
alone risk as compared to A.
Option C is not correct. Corporate risk is measured by the correlation of the project's
cash flows relative to the firm's existing projects. Higher, the correlation, higher the
corporate risk. Since, project B has higher correlation (.80), it has more corporate risk
as compared to project A.
Comparative advantage is an economic law referring to the ability of an individual or
group to carry out a specific economic activity at a lower relative opportunity cost or
autarky price in comparison to other economic actors. In order to determine if
comparative advantages exist between the two workers, we have to figure out the
opportunity cost of making one unit of one of the items. The worker who has the
lowest opportunity costs of producing one unit of product has the comparative
advantage and thus must specialize in performing the task.
Comparative Experiments is testing two variables by designing a experiment.This can
be designed to collect data on different/objects/events or collect data under different
condition to test the hypothesis.
It contains a testable hypothesis where we have at least one independent variable and
one dependent variables and a control group. These experiments are usually designed
to test the relationships between independent and dependent variable. Data can be
collected here by both quantitatively and/or qualitatively to draw conclusions and can
support/reject the hypothesis.
Once the results are received of the comparative experiment, the scientist can
compare the difference in the dependent variable C for each (independent variable)
treatment, concluding either one (independent variable) treatment is more effective
than the other or that both independent variable(treatments) have the same
effectiveness.
(c)
Population - The director of market research wants to conduct the study of the time
spent on shopping for clothing by the working women of the area, working women of
the area are the population of interest.
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide

Sample – The working women from the metropolitan area surveyed for the research.
b. Develop a first draft of the questionnaire needed in a(a) by writing three categorical
questions and three numerical questions that you feel would be appropriate for this
survey
Summary key points of methods to follow determining the caloric content listed in
nutritional labels of packaged food items. Under FDA:
· The nutrients records mark might be put along with the solving list and the
call and address (call and cope with of the manufacturer, packer, or merchant)
at the pdp. these 3 name articulations likewise is probably placed on the
statistics board (the mark board neighboring and to at least one aspect of the
pdp, or, if there is insufficient space on the contiguous board, on the following
nearby board to at least one facet). on bundles with missing sector on the pdp
and information board, the vitamins statistics call is probably put on
anotherBoard that can be visible via the purchaser. 21 cfr 101.2(b) and (e) and
one zero one.9(i)
· Even if utilizing the obvious presentation, the sustenance records ought to be
activate in a crate. 21 cfr one zero one.9(d)(1)(i)
· The call is probably set over the crate that encases the nourishment statistics.
21 cfr one zero one.9(c) and (d)
· There's no necessity that any records, aside from the net quantity of
substance and articulation of personality, be printed similar to the base of the
package deal. anyhow, fda urges manufacturers toMake progress towards
consistency of advent of nourishment facts on the lookout and to position the
vitamins records name with the aim that it is promptly perceptible and
intelligible to the shopper at the purpose of procurement
· The vertical association might be damaged in each of the accompanying
ways: (1) state of affairs of the connection with one facet of the board as
proven in the model in 21 cfr a hundred and one.nine(d)(11) or (2) all nutrients
and minerals that are recorded willfully (i.e., after iron)Might be moved to the
top right of the board alongside the remark. 21 cfr one zero one.9(d)(eleven)
· The nutrition facts mark (a model is represented in segment 7 l2) is needed
on most food bundles named. the delineation shows fda's typeface and fashion
to assist assure meaningfulness and obviousness. no longer those sort info are
required. the obligatory type particulars are recorded in 21 cfr one zero
one.9(d). in evaluation to the illustrative models on this route, (1) any readable
kind fashion is probablyUtilized, not without a doubt helvetica, (2) the
heading nutrients records need to be the most important kind size within the
nourishment mark (i.e., it must be larger than 8-factor, but should not be
thirteen-point) and must increase the width of the nutrition facts box, and (3)
there is no unique thickness wanted for the three bars that exclusive the focal
segments of the sustenance ¬ call. 21 cfr 101.9(a) and 21 cfr one zero
one.nine(a)(1)
b. Develop a first draft of the questionnaire needed in a(a) by writing three categorical
questions and three numerical questions that you feel would be appropriate for this
survey
Summary key points of methods to follow determining the caloric content listed in
nutritional labels of packaged food items. Under FDA:
· The nutrients records mark might be put along with the solving list and the
call and address (call and cope with of the manufacturer, packer, or merchant)
at the pdp. these 3 name articulations likewise is probably placed on the
statistics board (the mark board neighboring and to at least one aspect of the
pdp, or, if there is insufficient space on the contiguous board, on the following
nearby board to at least one facet). on bundles with missing sector on the pdp
and information board, the vitamins statistics call is probably put on
anotherBoard that can be visible via the purchaser. 21 cfr 101.2(b) and (e) and
one zero one.9(i)
· Even if utilizing the obvious presentation, the sustenance records ought to be
activate in a crate. 21 cfr one zero one.9(d)(1)(i)
· The call is probably set over the crate that encases the nourishment statistics.
21 cfr one zero one.9(c) and (d)
· There's no necessity that any records, aside from the net quantity of
substance and articulation of personality, be printed similar to the base of the
package deal. anyhow, fda urges manufacturers toMake progress towards
consistency of advent of nourishment facts on the lookout and to position the
vitamins records name with the aim that it is promptly perceptible and
intelligible to the shopper at the purpose of procurement
· The vertical association might be damaged in each of the accompanying
ways: (1) state of affairs of the connection with one facet of the board as
proven in the model in 21 cfr a hundred and one.nine(d)(11) or (2) all nutrients
and minerals that are recorded willfully (i.e., after iron)Might be moved to the
top right of the board alongside the remark. 21 cfr one zero one.9(d)(eleven)
· The nutrition facts mark (a model is represented in segment 7 l2) is needed
on most food bundles named. the delineation shows fda's typeface and fashion
to assist assure meaningfulness and obviousness. no longer those sort info are
required. the obligatory type particulars are recorded in 21 cfr one zero
one.9(d). in evaluation to the illustrative models on this route, (1) any readable
kind fashion is probablyUtilized, not without a doubt helvetica, (2) the
heading nutrients records need to be the most important kind size within the
nourishment mark (i.e., it must be larger than 8-factor, but should not be
thirteen-point) and must increase the width of the nutrition facts box, and (3)
there is no unique thickness wanted for the three bars that exclusive the focal
segments of the sustenance ¬ call. 21 cfr 101.9(a) and 21 cfr one zero
one.nine(a)(1)
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

· FDA considers facts that is required or allowed within the nutrients
information nameThat is on the front name or elsewhere on the package deal
outside the nutrients records mark to be a nutrient content declare (ncc). in
such cases, the package mark should consent to the suggestions for
complement content material cases. see the ncc phase and appendices an and b
of this document for greater data. 21 cfr one hundred and one.· In option to the
dietary supplements appeared on the call in location 7 l2 producers can also
upload calories from immersed fats, polyunsaturated fats, monounsaturated
fats, potassium, solvent andInsoluble fiber, sugar liquor, different carb,
vitamins and minerals for which reference each day intake (rdi's) were
installation, or the percent of nutrient a this is to be had as beta-carotene. 21
cfr one hundred and one.nine(c)
· Best the ones supplements recorded in fda's sustenance recommendations, as
required or intentional segments of the nourishment call, is probably
remembered for the nutrients facts mark. 21 cfr a hundred and one.nine(c)
· In option to the supplements appeared on the instance marks in this course,
REFERENCES:
1. Partnership for Public Service (November 2012). "The state of the FDA
workforce" (PDF). The Washington Post. Archived (PDF) from the original on
July 16, 2017. Retrieved May 12, 2017.
2. ^ Genomeweb (December 2019). "House Subcommittee Bill Proposes 7
Percent NIH Budget Increase". Staff Reporter. Archived from the original on
January 28, 2021. Retrieved January 23, 2020.
3. ^ "Animal Food & Feeds". Food and Drug Administration. Archived from the
original on March 22, 2015. Retrieved March 14, 2015.
4. ^ Kaplan, Sheila (January 20, 2021). "Hahn Leaves F.D.A.; Woodcock Named
Acting Commissioner". The New York Times. Archived January 22, 2021, at
the Wayback Machine
5. ^ Jump up to: a b "White Oak Campus Information". Food and Drug
Administration. February 9, 2011. Archived from the original on April 21,
2016. Retrieved May 12, 2017.
6. ^ Jump up to: a b "FDA Overview" (PDF). Food and Drug Administration.
Archived (PDF) from the original on November 1, 2013. Retrieved August 30,
2012.
7. ^ "FDA's International Posts: Improving the Safety of Imported Food and
Medical Products". Food and Drug Administration. Archived from the original
on August 10, 2010. Retrieved April 10, 2010.
8. ^ "About the FDA Organization Charts". Food and Drug Administration.
August 29, 2014. Archived from the original on July 22, 2015. Retrieved July
19, 2015. FDA is an agency within the Department of Health and Human
Services and consists of nine Centers and Offices, which are listed on the menu
to the left.
information nameThat is on the front name or elsewhere on the package deal
outside the nutrients records mark to be a nutrient content declare (ncc). in
such cases, the package mark should consent to the suggestions for
complement content material cases. see the ncc phase and appendices an and b
of this document for greater data. 21 cfr one hundred and one.· In option to the
dietary supplements appeared on the call in location 7 l2 producers can also
upload calories from immersed fats, polyunsaturated fats, monounsaturated
fats, potassium, solvent andInsoluble fiber, sugar liquor, different carb,
vitamins and minerals for which reference each day intake (rdi's) were
installation, or the percent of nutrient a this is to be had as beta-carotene. 21
cfr one hundred and one.nine(c)
· Best the ones supplements recorded in fda's sustenance recommendations, as
required or intentional segments of the nourishment call, is probably
remembered for the nutrients facts mark. 21 cfr a hundred and one.nine(c)
· In option to the supplements appeared on the instance marks in this course,
REFERENCES:
1. Partnership for Public Service (November 2012). "The state of the FDA
workforce" (PDF). The Washington Post. Archived (PDF) from the original on
July 16, 2017. Retrieved May 12, 2017.
2. ^ Genomeweb (December 2019). "House Subcommittee Bill Proposes 7
Percent NIH Budget Increase". Staff Reporter. Archived from the original on
January 28, 2021. Retrieved January 23, 2020.
3. ^ "Animal Food & Feeds". Food and Drug Administration. Archived from the
original on March 22, 2015. Retrieved March 14, 2015.
4. ^ Kaplan, Sheila (January 20, 2021). "Hahn Leaves F.D.A.; Woodcock Named
Acting Commissioner". The New York Times. Archived January 22, 2021, at
the Wayback Machine
5. ^ Jump up to: a b "White Oak Campus Information". Food and Drug
Administration. February 9, 2011. Archived from the original on April 21,
2016. Retrieved May 12, 2017.
6. ^ Jump up to: a b "FDA Overview" (PDF). Food and Drug Administration.
Archived (PDF) from the original on November 1, 2013. Retrieved August 30,
2012.
7. ^ "FDA's International Posts: Improving the Safety of Imported Food and
Medical Products". Food and Drug Administration. Archived from the original
on August 10, 2010. Retrieved April 10, 2010.
8. ^ "About the FDA Organization Charts". Food and Drug Administration.
August 29, 2014. Archived from the original on July 22, 2015. Retrieved July
19, 2015. FDA is an agency within the Department of Health and Human
Services and consists of nine Centers and Offices, which are listed on the menu
to the left.

9. ^ "Office of Operations Organization". Organization Charts. Food and Drug
Administration. April 15, 2015. Archived from the original on July 22, 2015.
Retrieved July 19, 2015.
10. ^ "Providing Easy Public Ac12cess to Prescription Drug, Over-the-Counter
Drug, and Biological Product Labeling". FDA Voice (blog). August 18, 2014.
footer. Archived from the original on July 22, 2015. Retrieved July 19, 2015.
11. ^ Taylor, Nick Paul (March 10, 2014). "FDA informatics chief: NGS is an
immediate priority for the agency". Regulatory. FierceBiotechIT. Archived
from the original on July 23, 2015. Retrieved July 19, 2015.
12. ^ "Buildings and Facilities". U.S. Food and Drug Administration. April 27,
2020. Archived from the original on April 21, 2020. Retrieved October 3,
2020.
13. ^ Coordinates of FDA Headquarters at White Oak, Maryland: 39°02′07″N
76°58′59″W
14. ^ Commissioner, Office of the (February 8, 2019). "White Oak Campus
Information". FDA. Archived from the original on December 14, 2019.
Retrieved April 2, 2020.
15. ^ "FDA White Oak Master Plan". National Capital Planning Commission.
Archived from the original on October 9, 2020. Retrieved October 3, 2020.
Administration. April 15, 2015. Archived from the original on July 22, 2015.
Retrieved July 19, 2015.
10. ^ "Providing Easy Public Ac12cess to Prescription Drug, Over-the-Counter
Drug, and Biological Product Labeling". FDA Voice (blog). August 18, 2014.
footer. Archived from the original on July 22, 2015. Retrieved July 19, 2015.
11. ^ Taylor, Nick Paul (March 10, 2014). "FDA informatics chief: NGS is an
immediate priority for the agency". Regulatory. FierceBiotechIT. Archived
from the original on July 23, 2015. Retrieved July 19, 2015.
12. ^ "Buildings and Facilities". U.S. Food and Drug Administration. April 27,
2020. Archived from the original on April 21, 2020. Retrieved October 3,
2020.
13. ^ Coordinates of FDA Headquarters at White Oak, Maryland: 39°02′07″N
76°58′59″W
14. ^ Commissioner, Office of the (February 8, 2019). "White Oak Campus
Information". FDA. Archived from the original on December 14, 2019.
Retrieved April 2, 2020.
15. ^ "FDA White Oak Master Plan". National Capital Planning Commission.
Archived from the original on October 9, 2020. Retrieved October 3, 2020.
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide
1 out of 6
Related Documents
Your All-in-One AI-Powered Toolkit for Academic Success.
+13062052269
info@desklib.com
Available 24*7 on WhatsApp / Email
![[object Object]](/_next/static/media/star-bottom.7253800d.svg)
Unlock your academic potential
Copyright © 2020–2026 A2Z Services. All Rights Reserved. Developed and managed by ZUCOL.




