Analyzing the US Controversy of Embryonic Stem Cell Research
VerifiedAdded on 2023/04/22
|7
|1662
|289
Essay
AI Summary
This essay examines the controversy surrounding the use of embryonic stem cells (ESCs) in the United States, focusing on the ethical and clinical debates. It begins by outlining the history of ESC research in the US, including the impact of presidential executive orders and legislative bans on federal funding. The essay then presents arguments for and against ESC use, highlighting the therapeutic potential of ESCs in regenerative medicine and drug development, while also addressing concerns about the destruction of human embryos and the violation of human dignity. Alternative approaches like adult embryonic stem cells are also considered. The essay concludes that while ESCs hold significant promise, ethical considerations remain a major obstacle to their widespread use, necessitating further research into alternative stem cell sources.

Running head: EMBRYONIC STEM CELL (ESC)
U.S Controversy of Embryonic Stem Cell (ESC)
Name of the Student
Name of the University
Author Note
U.S Controversy of Embryonic Stem Cell (ESC)
Name of the Student
Name of the University
Author Note
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

1
EMBRYONIC STEM CELL (ESC)
Stem cells are a broad class of cells that has potential for prolong self-renewal and
promising potential to differentiate into other types of cell line. Human Embryonic Stem
(HES) cells are extracted from the fertilized human egg before it is implemented into the
womb. The recently published research articles have highlighted the cells extracted from the
human embryos generated by in-vitro fertilization to the clinical purpose is useful in
generating pluripotent human ES cells (Tachibana et al., 2013). The following essay aims to
highlight the controversy related to use of the embryonic stem cells in the United States. The
essay will highlight both the positive and the negative sides behind the use of the human
ESCs in clinical practice.
The concept of the embryonic stem cell (hESC) came into active consideration in the
United States during March 2009 when the then President Barak Obama issued an executive
order that intended to bolster hESC research in the U.S. However, this announcement failed
to derive comprehensive federal support and funding for the hESC and evolving federal
policies further stymied the overall research efforts (King & Perrin, 2014). The former U.S
president Clinton signed a law in 1995 banning the federal funding of the research under the
rationale that it would destroy the human embryos and this barred researchers of the US
National Institutes of Health (NIH) grants from drafting new hESC lines (King & Perrin,
2014). The US Congress renews this ban every year and in 2001, the Bush administration
restricted the total number of the hESC ine that could be used in federally funded research,
only 21 viable lines were accepted. The executive order to the President Barak Obama over-
turned these constraints generated in the Bush era and thereby helping in proper
channelization of the U.S funding. However, the overall process behind the use of the hESC
is time-consuming and as it requires informed consent form the donor as per the NIH
principle (Nature Cell Biology, 2010).
EMBRYONIC STEM CELL (ESC)
Stem cells are a broad class of cells that has potential for prolong self-renewal and
promising potential to differentiate into other types of cell line. Human Embryonic Stem
(HES) cells are extracted from the fertilized human egg before it is implemented into the
womb. The recently published research articles have highlighted the cells extracted from the
human embryos generated by in-vitro fertilization to the clinical purpose is useful in
generating pluripotent human ES cells (Tachibana et al., 2013). The following essay aims to
highlight the controversy related to use of the embryonic stem cells in the United States. The
essay will highlight both the positive and the negative sides behind the use of the human
ESCs in clinical practice.
The concept of the embryonic stem cell (hESC) came into active consideration in the
United States during March 2009 when the then President Barak Obama issued an executive
order that intended to bolster hESC research in the U.S. However, this announcement failed
to derive comprehensive federal support and funding for the hESC and evolving federal
policies further stymied the overall research efforts (King & Perrin, 2014). The former U.S
president Clinton signed a law in 1995 banning the federal funding of the research under the
rationale that it would destroy the human embryos and this barred researchers of the US
National Institutes of Health (NIH) grants from drafting new hESC lines (King & Perrin,
2014). The US Congress renews this ban every year and in 2001, the Bush administration
restricted the total number of the hESC ine that could be used in federally funded research,
only 21 viable lines were accepted. The executive order to the President Barak Obama over-
turned these constraints generated in the Bush era and thereby helping in proper
channelization of the U.S funding. However, the overall process behind the use of the hESC
is time-consuming and as it requires informed consent form the donor as per the NIH
principle (Nature Cell Biology, 2010).

2
EMBRYONIC STEM CELL (ESC)
The argument in favor of the can be summarized as the arguments extracted from
the “ethics of healing” which upholds the therapeutic imperative. The ethics of healing is
further supported by the ethical principle of beneficence, which promote maximum benefit
under the cost of the minimal harm (Gafni et al., 2013). According to Lee et al. (2013) hESC
can proliferate without any restricted limit and thus can lead to the generation of the any cell
type. It also allows unprecedented access to the tissues from the human body through their
pluripotent nature. Gafni et al. (2013) highlighted that the drugs are generally not tested
directly over the human heart cells due to the absence of the human cell lines. Instead the
researchers mainly depend on the animal models. However, there exist stringent differences
between the animal and the human heart models moreover, the drugs that entered the clinical
trial has been found to impose the adverse affect to human. Under this context it can be said
that the hESC derived heart cell lines provides an ideal medium to testing the efficacy of the
drugs in the clinical trial. The drugs, which are successful in passing through the trial, will
have minimal chance of casting an adverse affect to human health. Such testing is not limited
t heart cells but to any type of cells, which is difficult to obtain from other, sources (Chong et
al., 2014). The research conducted by Chong et al. (2014), human embryonic stem cell
derived cardiomyoctes act as an effective source of model for cardiovascular drug trial.
Human ES cells also have comprehensive potential to provide innumerable amount of
the tissue for conducting successful transplantation in order to treat a wide range of the
degenerative disease. Some of the important human diseases are caused by the death or
subsequent dysfunction of the more than one fell types like the insulin-producing cells in
diabetes or the dopaminergic neurons in the neuro-degenerative disease like Parkinson. The
replacement of this fault, disease producing cells offer a life-long cure for debilitating
diseases (Lindvall, 2013).
EMBRYONIC STEM CELL (ESC)
The argument in favor of the can be summarized as the arguments extracted from
the “ethics of healing” which upholds the therapeutic imperative. The ethics of healing is
further supported by the ethical principle of beneficence, which promote maximum benefit
under the cost of the minimal harm (Gafni et al., 2013). According to Lee et al. (2013) hESC
can proliferate without any restricted limit and thus can lead to the generation of the any cell
type. It also allows unprecedented access to the tissues from the human body through their
pluripotent nature. Gafni et al. (2013) highlighted that the drugs are generally not tested
directly over the human heart cells due to the absence of the human cell lines. Instead the
researchers mainly depend on the animal models. However, there exist stringent differences
between the animal and the human heart models moreover, the drugs that entered the clinical
trial has been found to impose the adverse affect to human. Under this context it can be said
that the hESC derived heart cell lines provides an ideal medium to testing the efficacy of the
drugs in the clinical trial. The drugs, which are successful in passing through the trial, will
have minimal chance of casting an adverse affect to human health. Such testing is not limited
t heart cells but to any type of cells, which is difficult to obtain from other, sources (Chong et
al., 2014). The research conducted by Chong et al. (2014), human embryonic stem cell
derived cardiomyoctes act as an effective source of model for cardiovascular drug trial.
Human ES cells also have comprehensive potential to provide innumerable amount of
the tissue for conducting successful transplantation in order to treat a wide range of the
degenerative disease. Some of the important human diseases are caused by the death or
subsequent dysfunction of the more than one fell types like the insulin-producing cells in
diabetes or the dopaminergic neurons in the neuro-degenerative disease like Parkinson. The
replacement of this fault, disease producing cells offer a life-long cure for debilitating
diseases (Lindvall, 2013).
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide
3
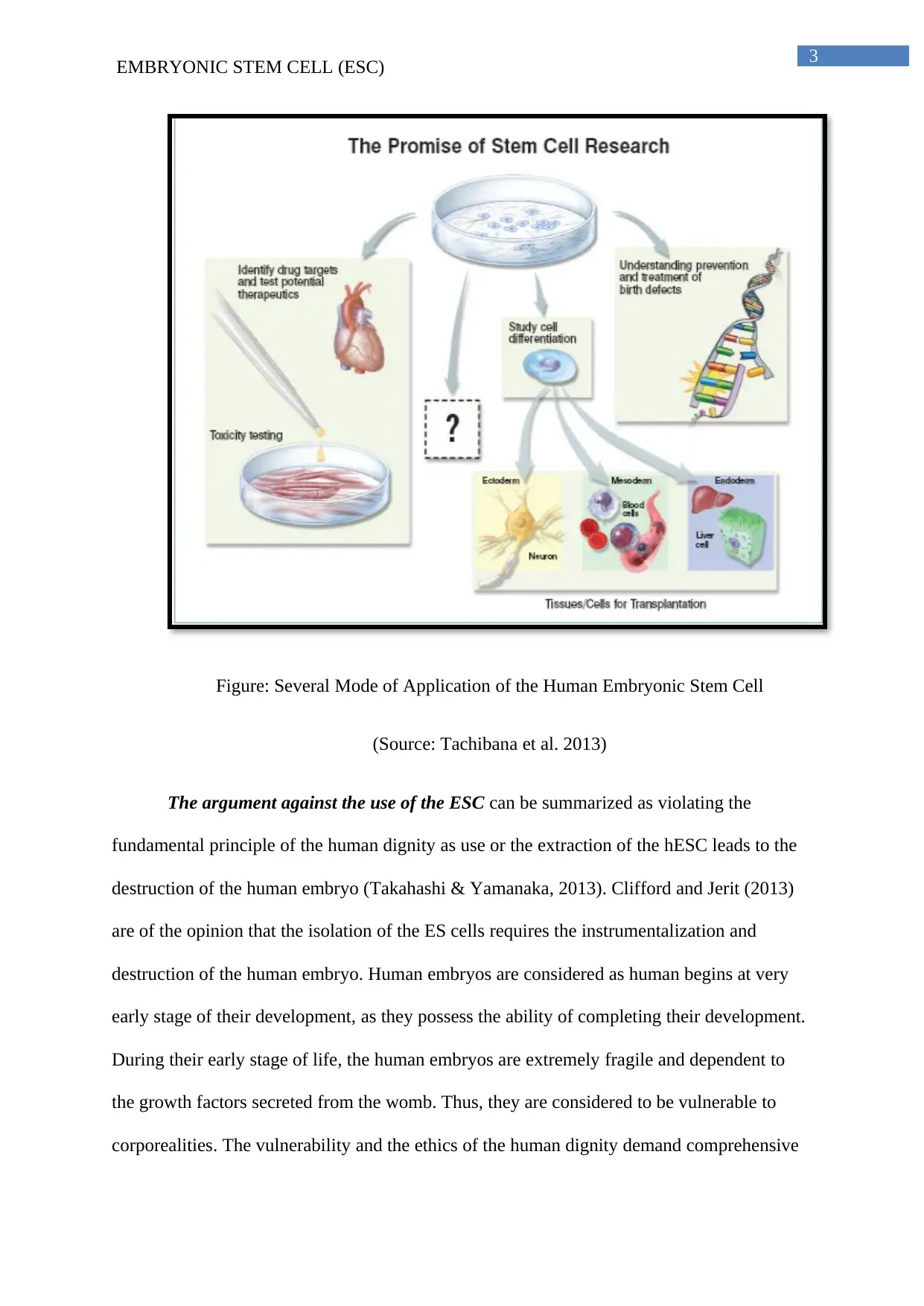
EMBRYONIC STEM CELL (ESC)
Figure: Several Mode of Application of the Human Embryonic Stem Cell
(Source: Tachibana et al. 2013)
The argument against the use of the ESC can be summarized as violating the
fundamental principle of the human dignity as use or the extraction of the hESC leads to the
destruction of the human embryo (Takahashi & Yamanaka, 2013). Clifford and Jerit (2013)
are of the opinion that the isolation of the ES cells requires the instrumentalization and
destruction of the human embryo. Human embryos are considered as human begins at very
early stage of their development, as they possess the ability of completing their development.
During their early stage of life, the human embryos are extremely fragile and dependent to
the growth factors secreted from the womb. Thus, they are considered to be vulnerable to
corporealities. The vulnerability and the ethics of the human dignity demand comprehensive
EMBRYONIC STEM CELL (ESC)
Figure: Several Mode of Application of the Human Embryonic Stem Cell
(Source: Tachibana et al. 2013)
The argument against the use of the ESC can be summarized as violating the
fundamental principle of the human dignity as use or the extraction of the hESC leads to the
destruction of the human embryo (Takahashi & Yamanaka, 2013). Clifford and Jerit (2013)
are of the opinion that the isolation of the ES cells requires the instrumentalization and
destruction of the human embryo. Human embryos are considered as human begins at very
early stage of their development, as they possess the ability of completing their development.
During their early stage of life, the human embryos are extremely fragile and dependent to
the growth factors secreted from the womb. Thus, they are considered to be vulnerable to
corporealities. The vulnerability and the ethics of the human dignity demand comprehensive
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

4
EMBRYONIC STEM CELL (ESC)
protection of the embryo’s corporeal integrity. Use of the hESCs inflicts controversy under
the ethical grounds of the non-maleficence avoidance of harm. In order to overcome the
ethical principle of non-maleficence, the proposal is given in favor of the adult embryonic
stem cell, which does not require the destruction of the human embryos. Moreover, adult
embryonic stem cell is also easily accessible like that of the human embryonic stem cell and
thereby creating further provision behind the effective use of the adult embryonic cell line for
the clinical research (Takahashi & Yamanaka, 2013).
Thus from the above discussion, it can be concluded that there are several benefits
behind the use of the human embryonic stem cells. The pluripotent nature of the human
embryonic stem cells confer them the power to regenerate into ay cell lines, tissues and
subsequent development of organ. Ability to propagate into any cell-line helps human
embryonic stem cell to act a source of model for the clinical trails of drugs. Moreover, it also
opens the new gateway towards the effective organ transplant. in 2009 March, then U.S
president Barak Obama issued an executive order that promoted further research of human
embryonic stem-cell under proper funding. However, there is still certain ethical obligation
behind the comprehensive use of the hESC. hESC breaches the ethical issue of the non-
maleficence as extraction of the hESC might lead to certain damage of the developing human
embryo. Further research proposal is undertaken behind the use of the adult embryonic stem
cells in order to avoid the ethical complications.
EMBRYONIC STEM CELL (ESC)
protection of the embryo’s corporeal integrity. Use of the hESCs inflicts controversy under
the ethical grounds of the non-maleficence avoidance of harm. In order to overcome the
ethical principle of non-maleficence, the proposal is given in favor of the adult embryonic
stem cell, which does not require the destruction of the human embryos. Moreover, adult
embryonic stem cell is also easily accessible like that of the human embryonic stem cell and
thereby creating further provision behind the effective use of the adult embryonic cell line for
the clinical research (Takahashi & Yamanaka, 2013).
Thus from the above discussion, it can be concluded that there are several benefits
behind the use of the human embryonic stem cells. The pluripotent nature of the human
embryonic stem cells confer them the power to regenerate into ay cell lines, tissues and
subsequent development of organ. Ability to propagate into any cell-line helps human
embryonic stem cell to act a source of model for the clinical trails of drugs. Moreover, it also
opens the new gateway towards the effective organ transplant. in 2009 March, then U.S
president Barak Obama issued an executive order that promoted further research of human
embryonic stem-cell under proper funding. However, there is still certain ethical obligation
behind the comprehensive use of the hESC. hESC breaches the ethical issue of the non-
maleficence as extraction of the hESC might lead to certain damage of the developing human
embryo. Further research proposal is undertaken behind the use of the adult embryonic stem
cells in order to avoid the ethical complications.

5
EMBRYONIC STEM CELL (ESC)
References
Chong, J. J., Yang, X., Don, C. W., Minami, E., Liu, Y. W., Weyers, J. J., ... & Gantz, J. A.
(2014). Human embryonic-stem-cell-derived cardiomyocytes regenerate non-human
primate hearts. Nature, 510(7504), 273.
Clifford, S., & Jerit, J. (2013). How words do the work of politics: Moral foundations theory
and the debate over stem cell research. The Journal of Politics, 75(3), 659-671.
Gafni, O., Weinberger, L., Mansour, A. A., Manor, Y. S., Chomsky, E., Ben-Yosef, D., ... &
Rais, Y. (2013). Derivation of novel human ground state naive pluripotent stem
cells. Nature, 504(7479), 282.
King, N. M., & Perrin, J. (2014). Ethical issues in stem cell research and therapy. Stem cell
research & therapy, 5(4), 85.
Lee, A. S., Tang, C., Rao, M. S., Weissman, I. L., & Wu, J. C. (2013). Tumorigenicity as a
clinical hurdle for pluripotent stem cell therapies. Nature medicine, 19(8), 998.
Lindvall, O. (2013). Developing dopaminergic cell therapy for Parkinson's disease—give up
or move forward?. Movement Disorders, 28(3), 268-273.
Nature Cell biology. (2010). Human embryonic stem cell research in the US: time for
change?. Volume 12 (627). Access date: 25th Feb 2019. Retrieved from:
https://www.nature.com/articles/ncb0710-627
Tachibana, M., Amato, P., Sparman, M., Gutierrez, N. M., Tippner-Hedges, R., Ma, H., ... &
Masterson, K. (2013). Human embryonic stem cells derived by somatic cell nuclear
transfer. Cell, 153(6), 1228-1238.
EMBRYONIC STEM CELL (ESC)
References
Chong, J. J., Yang, X., Don, C. W., Minami, E., Liu, Y. W., Weyers, J. J., ... & Gantz, J. A.
(2014). Human embryonic-stem-cell-derived cardiomyocytes regenerate non-human
primate hearts. Nature, 510(7504), 273.
Clifford, S., & Jerit, J. (2013). How words do the work of politics: Moral foundations theory
and the debate over stem cell research. The Journal of Politics, 75(3), 659-671.
Gafni, O., Weinberger, L., Mansour, A. A., Manor, Y. S., Chomsky, E., Ben-Yosef, D., ... &
Rais, Y. (2013). Derivation of novel human ground state naive pluripotent stem
cells. Nature, 504(7479), 282.
King, N. M., & Perrin, J. (2014). Ethical issues in stem cell research and therapy. Stem cell
research & therapy, 5(4), 85.
Lee, A. S., Tang, C., Rao, M. S., Weissman, I. L., & Wu, J. C. (2013). Tumorigenicity as a
clinical hurdle for pluripotent stem cell therapies. Nature medicine, 19(8), 998.
Lindvall, O. (2013). Developing dopaminergic cell therapy for Parkinson's disease—give up
or move forward?. Movement Disorders, 28(3), 268-273.
Nature Cell biology. (2010). Human embryonic stem cell research in the US: time for
change?. Volume 12 (627). Access date: 25th Feb 2019. Retrieved from:
https://www.nature.com/articles/ncb0710-627
Tachibana, M., Amato, P., Sparman, M., Gutierrez, N. M., Tippner-Hedges, R., Ma, H., ... &
Masterson, K. (2013). Human embryonic stem cells derived by somatic cell nuclear
transfer. Cell, 153(6), 1228-1238.
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide

6
EMBRYONIC STEM CELL (ESC)
Takahashi, K., & Yamanaka, S. (2013). Induced pluripotent stem cells in medicine and
biology. Development, 140(12), 2457-2461.
EMBRYONIC STEM CELL (ESC)
Takahashi, K., & Yamanaka, S. (2013). Induced pluripotent stem cells in medicine and
biology. Development, 140(12), 2457-2461.
1 out of 7
Your All-in-One AI-Powered Toolkit for Academic Success.
+13062052269
info@desklib.com
Available 24*7 on WhatsApp / Email
![[object Object]](/_next/static/media/star-bottom.7253800d.svg)
Unlock your academic potential
Copyright © 2020–2026 A2Z Services. All Rights Reserved. Developed and managed by ZUCOL.