U.S. FDA Requirements on Product Labeling for Public Safety
VerifiedAdded on 2021/04/17
|14
|1005
|107
Report
AI Summary
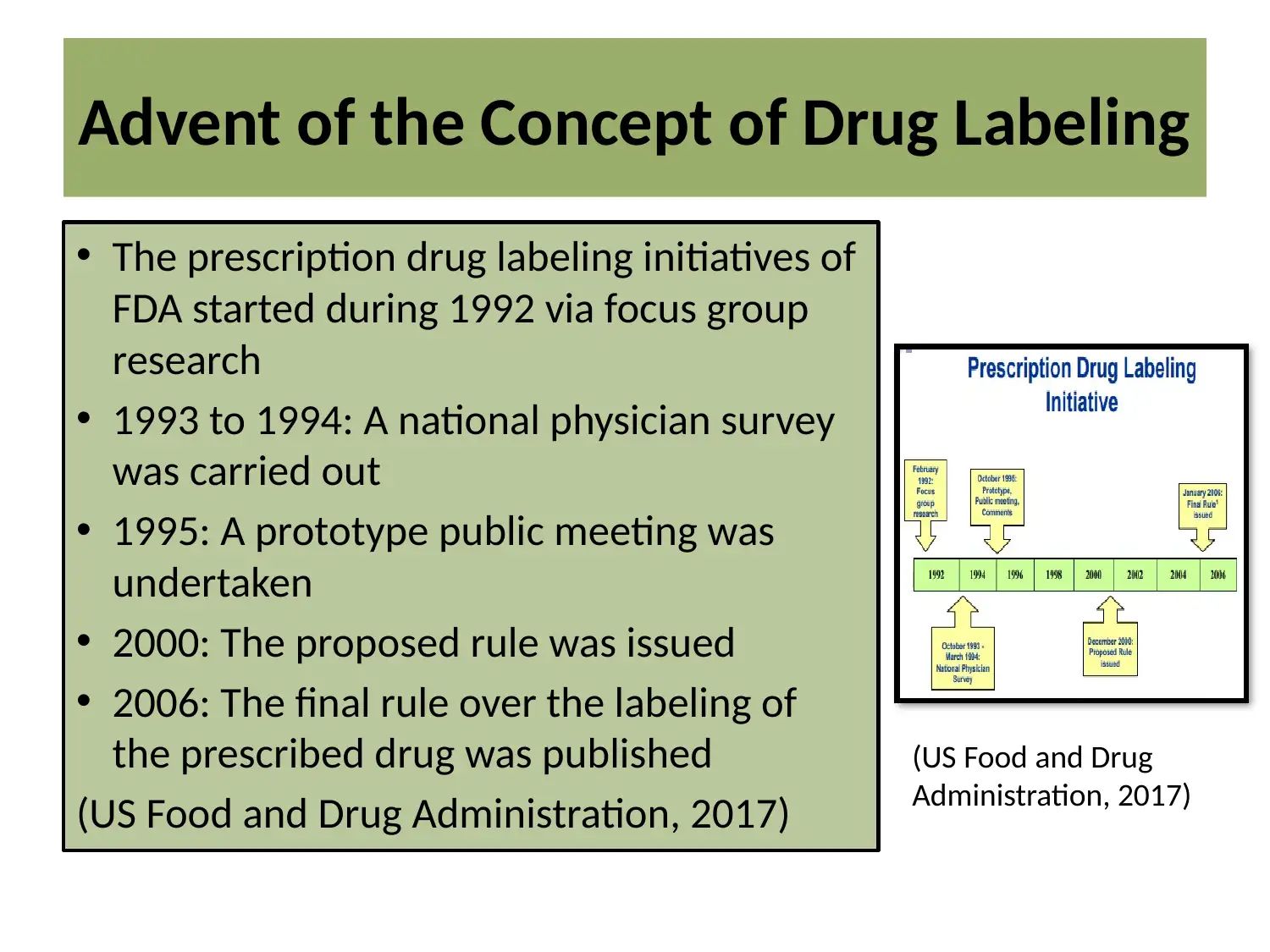
This report provides an overview of the U.S. Food and Drug Administration's (FDA) requirements for product labeling, focusing on its role in ensuring public safety. It details the two main types of drug labeling: over-the-counter (OTC) and prescription drugs, highlighting the evolution of FDA's labeling initiatives, starting from the 1990s. The report outlines the purpose of drug labeling, which is to provide healthcare professionals with essential information for appropriate drug prescription and to inform consumers. It also covers the key requirements of drug labeling, including statements on limitations, product names, warnings, and adverse reactions. Furthermore, the report discusses the limitations of current labeling practices, such as patient confusion, and explores the application of the FDA Label Database. The conclusion emphasizes the importance of drug labeling in preventing medication errors and promoting informed healthcare decisions, referencing relevant literature and databases.
1 out of 14














![[object Object]](/_next/static/media/star-bottom.7253800d.svg)