University Plant and Process Design Report - EGH463
VerifiedAdded on 2022/12/30
|7
|687
|43
Report
AI Summary
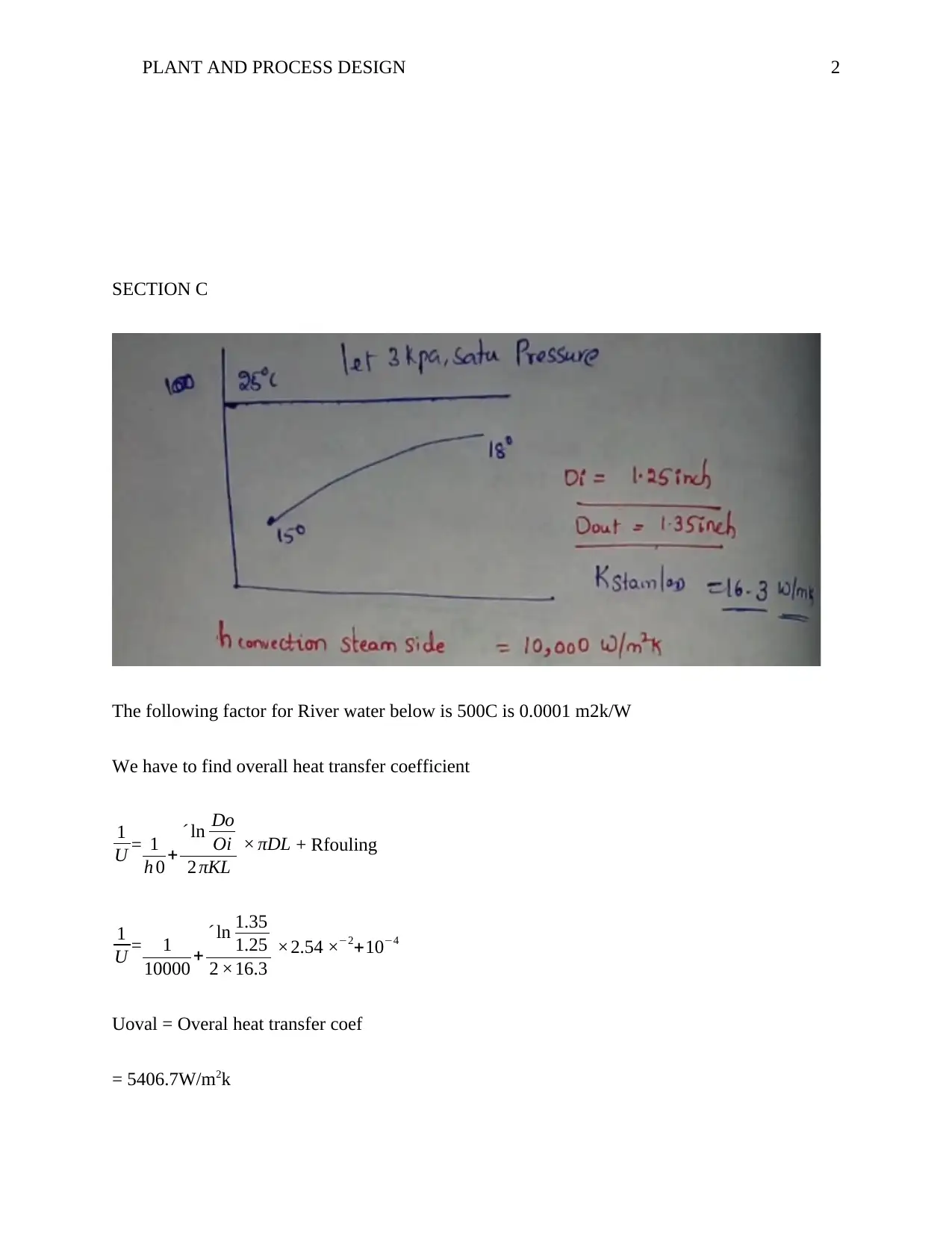
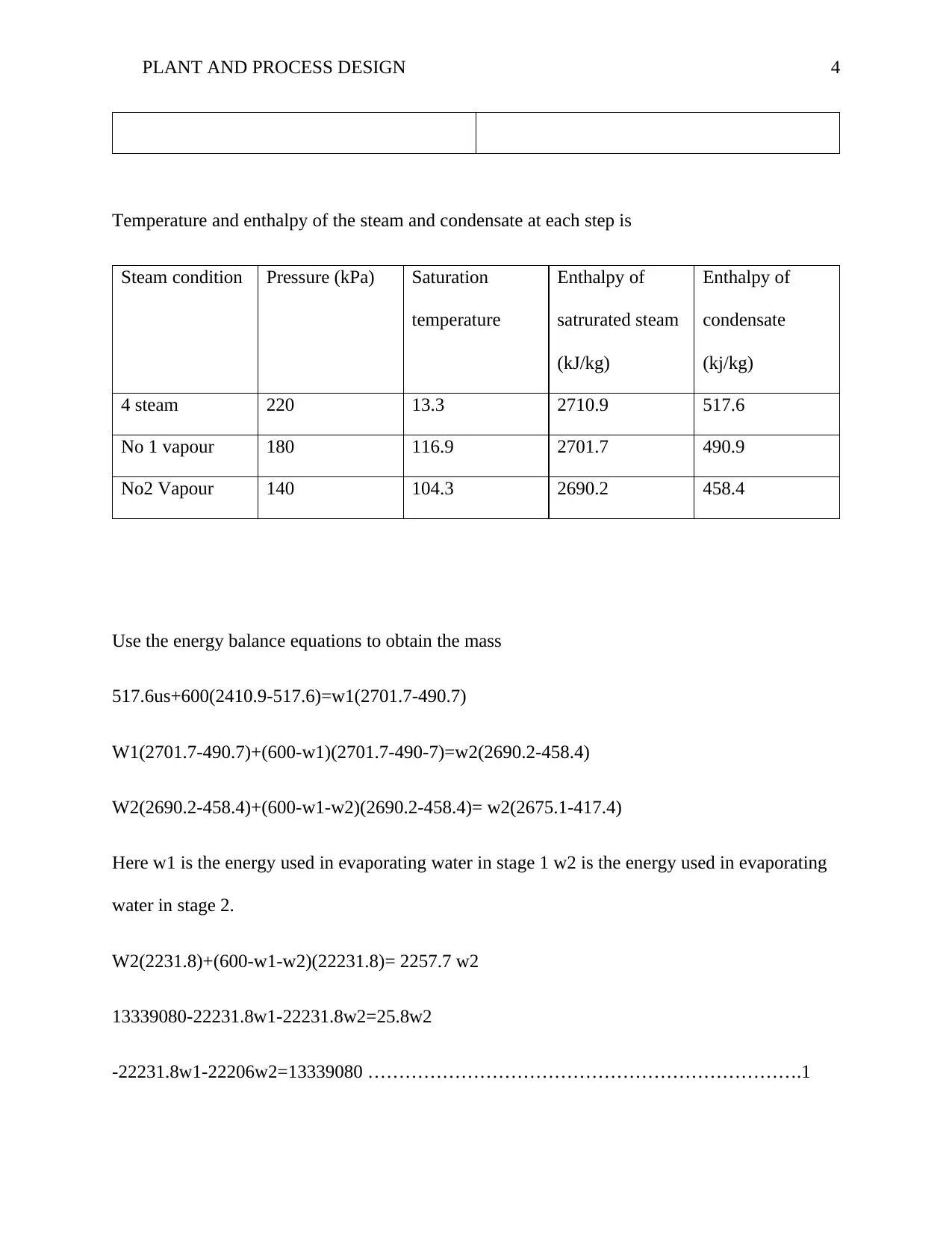
This report analyzes plant and process design, focusing on Section C's heat transfer coefficient calculation, and Section D's multi-stage evaporation process. It includes calculations for the overall heat transfer coefficient, mass flow rate, and energy balance equations for steam conditions. The report further addresses the quantity of water evaporated, and the minimum cooling water required for condensation, referencing relevant equations and data. The assignment also includes the P&ID diagram for IGCC schematic. The report concludes with a list of references, demonstrating a comprehensive understanding of plant design principles and their application in industrial processes. This assignment is a great example of how engineering principles are applied to solve real world problems.
1 out of 7







![[object Object]](/_next/static/media/star-bottom.7253800d.svg)