CBMS123 Essay: Small Molecules in Somatic Cell Reprogramming Analysis
VerifiedAdded on 2023/06/03
|13
|3068
|197
Essay
AI Summary
This essay delves into the significance of small molecules in somatic cell reprogramming, focusing on their role in the development of induced pluripotent stem cells (iPSCs). It explores the mechanisms by which these molecules influence cell fate, highlighting their potential in regenerative medicine, disease modeling, and drug discovery. The essay examines the benefits of small molecules, such as their structural diversity, rapid action, and ease of administration, while also acknowledging their limitations. It discusses the historical context of cell reprogramming, including the use of somatic cell nuclear transfer and cell fusion, and the challenges associated with these methods. Furthermore, the essay investigates the use of small molecules to replace transcription factors like Sox2, c-Myc, and Klf4, and their impact on iPSC generation efficiency. It concludes by emphasizing the transformative potential of small molecules in stem cell research, while acknowledging the need for addressing safety concerns and improving the efficiency and quality of reprogrammed cells for clinical applications. The essay also includes figures illustrating chemical techniques to stem cell biology and therapeutics and small molecule-regulated reprogramming.

Small molecules in somatic cell
reprogramming1
SMALL MOLECULES IN SOMATIC CELL REPROGRAMMING
Name:
Department:
School:
Date:
reprogramming1
SMALL MOLECULES IN SOMATIC CELL REPROGRAMMING
Name:
Department:
School:
Date:
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

Small molecules in somatic cell reprogramming 2
Small molecules in somatic cell reprogramming
Recent advancement in the arena of induced pluripotent stem cells (iPSCs) has disclosed
numerous entries for the therapeutics investigation. “IPSCs are the cells which are
reprogrammed from somatic cells using various transcription features”1. IPSCs have distinctive
properties of differentiation and self-renewal to numerous sorts of cell heredity. Therefore, one
can substitute the application of embryonic stem cells and may overcome the numerous moral
subjects concerning the usage of embryo in clinics and research. The molecular mechanism that
plays part in reprogramming to produce iPSCs from various forms of somatic cells bases
comprises an excess of fragments such as DNA adjusting mediators (DNA methyltransferases)
and miRNAs. IPSCs could be utilised in the study of molecular mechanism of many diseases.
IPSCs skill has been used in numerous infections for gene therapy and syndrome modelling. The
method offers paybacks over other similar technologies such as animal model. Numerous deadly
substances which are encountered by human may be assessed for harmfulness and effect by
using iPSCs. Therefore, iPSCs application in regenerative drug, disease modelling and
medication innovation are massive and ought to be evaluated in a more comprehensive manner2.
1 Kamao, H., Mandai, M., Okamoto, S., Sakai, N., Suga, A., Sugita, S., Kiryu, J. and
Takahashi, M., 2014. Characterization of human induced pluripotent stem cell-derived
retinal pigment epithelium cell sheets aiming for clinical application. Stem cell
reports, 2(2), pp.205-218.
2 Banito, A. and Gil, J., 2010. Induced pluripotent stem cells and senescence: learning the
biology to improve the technology. EMBO reports, 11(5), pp.353-359. Retrieved from:
http://embor.embopress.org/content/11/5/353.abstract, [Accessed on 25 October 2018].
Small molecules in somatic cell reprogramming
Recent advancement in the arena of induced pluripotent stem cells (iPSCs) has disclosed
numerous entries for the therapeutics investigation. “IPSCs are the cells which are
reprogrammed from somatic cells using various transcription features”1. IPSCs have distinctive
properties of differentiation and self-renewal to numerous sorts of cell heredity. Therefore, one
can substitute the application of embryonic stem cells and may overcome the numerous moral
subjects concerning the usage of embryo in clinics and research. The molecular mechanism that
plays part in reprogramming to produce iPSCs from various forms of somatic cells bases
comprises an excess of fragments such as DNA adjusting mediators (DNA methyltransferases)
and miRNAs. IPSCs could be utilised in the study of molecular mechanism of many diseases.
IPSCs skill has been used in numerous infections for gene therapy and syndrome modelling. The
method offers paybacks over other similar technologies such as animal model. Numerous deadly
substances which are encountered by human may be assessed for harmfulness and effect by
using iPSCs. Therefore, iPSCs application in regenerative drug, disease modelling and
medication innovation are massive and ought to be evaluated in a more comprehensive manner2.
1 Kamao, H., Mandai, M., Okamoto, S., Sakai, N., Suga, A., Sugita, S., Kiryu, J. and
Takahashi, M., 2014. Characterization of human induced pluripotent stem cell-derived
retinal pigment epithelium cell sheets aiming for clinical application. Stem cell
reports, 2(2), pp.205-218.
2 Banito, A. and Gil, J., 2010. Induced pluripotent stem cells and senescence: learning the
biology to improve the technology. EMBO reports, 11(5), pp.353-359. Retrieved from:
http://embor.embopress.org/content/11/5/353.abstract, [Accessed on 25 October 2018].

Small molecules in somatic cell reprogramming 3
In spite of the huge prospective of stem cells for simple clinical and research use, hitches
such as their difficulty and rare accessibility in managing their outcome require to be articulated
to completely accomplish their probable. The current accomplishment of cellular reprogramming
has facilitated the formation of iPSCs from more sufficient somatic cell types by distinct
hereditary elements. But, severe concern remains about the safety and efficiency of
contemporary genomic methods to cell reprogramming and orthodox philosophy system that are
applied for stem cell care. As balancing techniques, small fragments that target particular
signalling paths, epigenetic and other cellular procedures provide dominant approaches for
manipulation of cell fate to a required result3. An advancing figure of small particles have been
realized to uphold the self-renewal prospective of stem cell, to prompt lineage distinction and to
back reprograming by growing the reprogramming efficacy or by substituting hereditary
reprograming elements4. Additionally, systematic examinations of the effects of these substances
also offer novel biological intuitions. Here, the paper examines the accomplishments in the
3 Marks, H., Kalkan, T., Menafra, R., Denissov, S., Jones, K., Hofemeister, H., Nichols, J.,
Kranz, A., Stewart, A.F., Smith, A. and Stunnenberg, H.G., 2012. The transcriptional and
epigenomic foundations of ground state pluripotency. Cell, 149(3), pp.590-604. Retrieved from:
https://doi.org/10.1016/j.cell.2012.03.026, [Accessed on 25 October 2018].
4 Nie, B., Wang, H., Laurent, T. and Ding, S., 2012. Cellular reprogramming: a small molecule
perspective. Current opinion in cell biology, 24(6), pp.784-792. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S0955067412001354, [Accessed on 25
October 2018].
In spite of the huge prospective of stem cells for simple clinical and research use, hitches
such as their difficulty and rare accessibility in managing their outcome require to be articulated
to completely accomplish their probable. The current accomplishment of cellular reprogramming
has facilitated the formation of iPSCs from more sufficient somatic cell types by distinct
hereditary elements. But, severe concern remains about the safety and efficiency of
contemporary genomic methods to cell reprogramming and orthodox philosophy system that are
applied for stem cell care. As balancing techniques, small fragments that target particular
signalling paths, epigenetic and other cellular procedures provide dominant approaches for
manipulation of cell fate to a required result3. An advancing figure of small particles have been
realized to uphold the self-renewal prospective of stem cell, to prompt lineage distinction and to
back reprograming by growing the reprogramming efficacy or by substituting hereditary
reprograming elements4. Additionally, systematic examinations of the effects of these substances
also offer novel biological intuitions. Here, the paper examines the accomplishments in the
3 Marks, H., Kalkan, T., Menafra, R., Denissov, S., Jones, K., Hofemeister, H., Nichols, J.,
Kranz, A., Stewart, A.F., Smith, A. and Stunnenberg, H.G., 2012. The transcriptional and
epigenomic foundations of ground state pluripotency. Cell, 149(3), pp.590-604. Retrieved from:
https://doi.org/10.1016/j.cell.2012.03.026, [Accessed on 25 October 2018].
4 Nie, B., Wang, H., Laurent, T. and Ding, S., 2012. Cellular reprogramming: a small molecule
perspective. Current opinion in cell biology, 24(6), pp.784-792. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S0955067412001354, [Accessed on 25
October 2018].
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide

Small molecules in somatic cell reprogramming 4
upkeep of stem cell comprising the iPSCs reprogramming, with the focus on influence of trivial
molecules.
Small particles deliver a striking technique for addressing the challenge, as they give a
number of convincing benefits. First, the structural range that can be offered by artificial
chemistry permits the functional optimization of trivial fragments. Second, the biological effects
of trivial bits are generally rapid, revisable and dose-dependent, permitting exact regulation over
precise result by fine-tuning their combination and concentrations. Third, matched with the
genomic intervention, the comparative ease of the administration and handling of small
fragments make them more useful for in vivo and in vitro use, and for extra healing
advancement. But, small particles have their own drawbacks. Particularly, small molecules can
have more than one object. Additionally, unforeseen toxicity in vivo may inhibit the clinical use
of trivial molecules. But, the prospective of small bits to develop the arena of stem cell
investigation should not be underrated.
The reverse of differentiation and the PSCs generation from the somatic cell have riveted
scholar for decades. The primary research of somatic cell nuclear transfer discovered that a
somatic nucleus can be entirely reprogrammed into totipotent condition by elements from an
enucleated egg, continue to the generation of an whole organism or be applied a drive ESCs.
Despite many advances in SCNT to generate hESCs, the process remains technically hard, and in
the human body, there are moral apprehensions to usage of SCNT to form hESCs5. Another
5 Ying, Q.L., Wray, J., Nichols, J., Batlle-Morera, L., Doble, B., Woodgett, J., Cohen, P. and
Smith, A., 2008. The ground state of embryonic stem cell self-renewal. Nature, 453(7194),
p.519. Retrieved from: https://www.nature.com/articles/nature06968, [Accessed on 25 October
2018].
upkeep of stem cell comprising the iPSCs reprogramming, with the focus on influence of trivial
molecules.
Small particles deliver a striking technique for addressing the challenge, as they give a
number of convincing benefits. First, the structural range that can be offered by artificial
chemistry permits the functional optimization of trivial fragments. Second, the biological effects
of trivial bits are generally rapid, revisable and dose-dependent, permitting exact regulation over
precise result by fine-tuning their combination and concentrations. Third, matched with the
genomic intervention, the comparative ease of the administration and handling of small
fragments make them more useful for in vivo and in vitro use, and for extra healing
advancement. But, small particles have their own drawbacks. Particularly, small molecules can
have more than one object. Additionally, unforeseen toxicity in vivo may inhibit the clinical use
of trivial molecules. But, the prospective of small bits to develop the arena of stem cell
investigation should not be underrated.
The reverse of differentiation and the PSCs generation from the somatic cell have riveted
scholar for decades. The primary research of somatic cell nuclear transfer discovered that a
somatic nucleus can be entirely reprogrammed into totipotent condition by elements from an
enucleated egg, continue to the generation of an whole organism or be applied a drive ESCs.
Despite many advances in SCNT to generate hESCs, the process remains technically hard, and in
the human body, there are moral apprehensions to usage of SCNT to form hESCs5. Another
5 Ying, Q.L., Wray, J., Nichols, J., Batlle-Morera, L., Doble, B., Woodgett, J., Cohen, P. and
Smith, A., 2008. The ground state of embryonic stem cell self-renewal. Nature, 453(7194),
p.519. Retrieved from: https://www.nature.com/articles/nature06968, [Accessed on 25 October
2018].
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

Small molecules in somatic cell reprogramming 5
drawback of SCNT and cell fusion is to intercede reprogramming, they apply mostly undefined
cellular matters, which cause it to be hard to examine the fundamental mechanisms. Also, the
cell fusion between somatic cells and ESCs to create heterokaryons can also reprogram somatic
nuclei into the pluripotent conditions, even though this normally effects in the existence of the
additional arrays of chromosomes6.
Human iPSCs are alike to human ESCs in sense that they have the prospective to separate
into cells of all three germ films. Elderly patients with degenerative diseases, injuries, cancers
would benefit from the stem cell-based regenerative medical methods. The iPSCs applications is
potential in cell transplantation and stimulate the regenerative medicine of endogenous cells to
rebuild tissues, in vitro medicine screening and disease modelling.
Initially, the adult cells were induced into iPSCs over exogenous overexpression of the
transcription elements Oct 4, Sox, Klf4 and cMyc. But, the efficacy of this method is at a very
low phase, with about 0.1% of mouse fibroblast and 0.01% of human fibroblasts cell7. The slow
dynamics and low efficiency of this technique posed severe potential concerns for the generation
6 Jaenisch, R., 2012. Nuclear cloning and direct reprogramming: the long and the short path to
Stockholm. Cell Stem Cell, 11(6), pp.744-747. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S193459091200639X, [Accessed on 25
October 2018].
7 Li, R., Liang, J., Ni, S., Zhou, T., Qing, X., Li, H., He, W., Chen, J., Li, F., Zhuang, Q. and Qin,
B., 2010. A mesenchymal-to-epithelial transition initiates and is required for the nuclear
reprogramming of mouse fibroblasts. Cell stem cell, 7(1), pp.51-63. Retrieved from:
https://doi.org/10.1016/j.stem.2010.04.014, [Accessed on 25 October 2018].
drawback of SCNT and cell fusion is to intercede reprogramming, they apply mostly undefined
cellular matters, which cause it to be hard to examine the fundamental mechanisms. Also, the
cell fusion between somatic cells and ESCs to create heterokaryons can also reprogram somatic
nuclei into the pluripotent conditions, even though this normally effects in the existence of the
additional arrays of chromosomes6.
Human iPSCs are alike to human ESCs in sense that they have the prospective to separate
into cells of all three germ films. Elderly patients with degenerative diseases, injuries, cancers
would benefit from the stem cell-based regenerative medical methods. The iPSCs applications is
potential in cell transplantation and stimulate the regenerative medicine of endogenous cells to
rebuild tissues, in vitro medicine screening and disease modelling.
Initially, the adult cells were induced into iPSCs over exogenous overexpression of the
transcription elements Oct 4, Sox, Klf4 and cMyc. But, the efficacy of this method is at a very
low phase, with about 0.1% of mouse fibroblast and 0.01% of human fibroblasts cell7. The slow
dynamics and low efficiency of this technique posed severe potential concerns for the generation
6 Jaenisch, R., 2012. Nuclear cloning and direct reprogramming: the long and the short path to
Stockholm. Cell Stem Cell, 11(6), pp.744-747. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S193459091200639X, [Accessed on 25
October 2018].
7 Li, R., Liang, J., Ni, S., Zhou, T., Qing, X., Li, H., He, W., Chen, J., Li, F., Zhuang, Q. and Qin,
B., 2010. A mesenchymal-to-epithelial transition initiates and is required for the nuclear
reprogramming of mouse fibroblasts. Cell stem cell, 7(1), pp.51-63. Retrieved from:
https://doi.org/10.1016/j.stem.2010.04.014, [Accessed on 25 October 2018].

Small molecules in somatic cell reprogramming 6
of iPSCs. On the other hand, low iPSCs generation efficiency; there is some safety problem with
them regarding the overexpression of the fours aforementioned transcription factors comprising
genetic mutations, incomplete reprogramming, gene insertions, epigenetic changes and
immunogenicity8.
Small molecules that might substitute Sox2
The elements such as SB431542 and E-616452 are changing the growth factors- TGF-
beta inhibitors that can substitute Sox2 during the human and mouse iPSC generation. But, E-
616452 does not actually perform by inducing Sox2 expression in the targets cells. It rather
facilitates reprogramming through the Nanog transcription induction. Another TGF- inhibitor is
LY-364947 which can substitute Sox2 in a miPSC generation. In mouse fibroblasts,
purmorphamine, Shh, and oxysterol have been reported to unregulate the N-Myc, So2 and Bmil
expression9.
Small molecules that might replace c-Myc and Klf4
8 Maherali, N. and Hochedlinger, K., 2009. Tgfβ signal inhibition cooperates in the induction of
iPSCs and replaces Sox2 and cMyc. Current Biology, 19(20), pp.1718-1723. Retrieved from:
https://doi.org/10.1016/j.cub.2009.08.025, [Accessed on 25 October 2018].
9 Ichida, J.K., Blanchard, J., Lam, K., Son, E.Y., Chung, J.E., Egli, D., Loh, K.M., Carter, A.C.,
Di Giorgio, F.P., Koszka, K. and Huangfu, D., 2009. A small-molecule inhibitor of Tgf-β
signaling replaces Sox2 in reprogramming by inducing Nanog. Cell stem cell, 5(5), pp.491-503.
Retrieved from: https://www.sciencedirect.com/science/article/pii/S1934590909005086,
[Accessed on 25 October 2018].
of iPSCs. On the other hand, low iPSCs generation efficiency; there is some safety problem with
them regarding the overexpression of the fours aforementioned transcription factors comprising
genetic mutations, incomplete reprogramming, gene insertions, epigenetic changes and
immunogenicity8.
Small molecules that might substitute Sox2
The elements such as SB431542 and E-616452 are changing the growth factors- TGF-
beta inhibitors that can substitute Sox2 during the human and mouse iPSC generation. But, E-
616452 does not actually perform by inducing Sox2 expression in the targets cells. It rather
facilitates reprogramming through the Nanog transcription induction. Another TGF- inhibitor is
LY-364947 which can substitute Sox2 in a miPSC generation. In mouse fibroblasts,
purmorphamine, Shh, and oxysterol have been reported to unregulate the N-Myc, So2 and Bmil
expression9.
Small molecules that might replace c-Myc and Klf4
8 Maherali, N. and Hochedlinger, K., 2009. Tgfβ signal inhibition cooperates in the induction of
iPSCs and replaces Sox2 and cMyc. Current Biology, 19(20), pp.1718-1723. Retrieved from:
https://doi.org/10.1016/j.cub.2009.08.025, [Accessed on 25 October 2018].
9 Ichida, J.K., Blanchard, J., Lam, K., Son, E.Y., Chung, J.E., Egli, D., Loh, K.M., Carter, A.C.,
Di Giorgio, F.P., Koszka, K. and Huangfu, D., 2009. A small-molecule inhibitor of Tgf-β
signaling replaces Sox2 in reprogramming by inducing Nanog. Cell stem cell, 5(5), pp.491-503.
Retrieved from: https://www.sciencedirect.com/science/article/pii/S1934590909005086,
[Accessed on 25 October 2018].
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide

Small molecules in somatic cell reprogramming 7
Even though WNT signalling pathway regulators can advance the iPSCs generation
effectiveness, they can also substitute the c-Myc function. Moreover, numerous trivial molecules
can upsurge the iPSCs generation efficacy by replacing the c-Myc and Klf4 in the course of the
somatic cell reprogramming into iPSCs. Kenpaullone has been proposed to be an alternative for
Klf4 in mouse cells, even though the basic mechanism is a mystery10.
Small molecules that substitute Klf4, Sox2 and c-Myc
It is through the small molecules linking such as VC6T simplify miPSC generation by
minimizing some major blockades to the reprogramming course.
To improve the quality and efficiency of iPSCs induction, more exertions has been
applied in the advancement of novel iPSCs generation techniques through the application of
integration and non-integration recombinant viruses, episomal vectors, DNA expression vectors,
minicircle vectors and liposomal magnetofection. Non-DNA techniques comprise proteins,
mRNA fragments and numerous chemical agents have been tested and a chemical technique that
creates iPSCs appears to be the most hopeful methods.
Even though the human iPSCs using chemical merely has not been advanced yet, human
stem cell researched with small molecules are revealing extra details about the epigenetic
remodelling. Thus, hopefully, this research might relieve challenges about the kinetics,
10 Yoshida, Y., Takahashi, K., Okita, K., Ichisaka, T. and Yamanaka, S., 2009. Hypoxia enhances
the generation of induced pluripotent stem cells. Cell stem cell, 5(3), pp.237-241. Retrieved
from: https://www.cell.com/cell-stem-cell/abstract/S1934-5909(09)00385-3, [Accessed on 25
October 2018].
Even though WNT signalling pathway regulators can advance the iPSCs generation
effectiveness, they can also substitute the c-Myc function. Moreover, numerous trivial molecules
can upsurge the iPSCs generation efficacy by replacing the c-Myc and Klf4 in the course of the
somatic cell reprogramming into iPSCs. Kenpaullone has been proposed to be an alternative for
Klf4 in mouse cells, even though the basic mechanism is a mystery10.
Small molecules that substitute Klf4, Sox2 and c-Myc
It is through the small molecules linking such as VC6T simplify miPSC generation by
minimizing some major blockades to the reprogramming course.
To improve the quality and efficiency of iPSCs induction, more exertions has been
applied in the advancement of novel iPSCs generation techniques through the application of
integration and non-integration recombinant viruses, episomal vectors, DNA expression vectors,
minicircle vectors and liposomal magnetofection. Non-DNA techniques comprise proteins,
mRNA fragments and numerous chemical agents have been tested and a chemical technique that
creates iPSCs appears to be the most hopeful methods.
Even though the human iPSCs using chemical merely has not been advanced yet, human
stem cell researched with small molecules are revealing extra details about the epigenetic
remodelling. Thus, hopefully, this research might relieve challenges about the kinetics,
10 Yoshida, Y., Takahashi, K., Okita, K., Ichisaka, T. and Yamanaka, S., 2009. Hypoxia enhances
the generation of induced pluripotent stem cells. Cell stem cell, 5(3), pp.237-241. Retrieved
from: https://www.cell.com/cell-stem-cell/abstract/S1934-5909(09)00385-3, [Accessed on 25
October 2018].
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser
Small molecules in somatic cell reprogramming 8
specificity, safety and efficiency of generating human iPSCs and bring human iPSCs nearer to
effective clinical application.
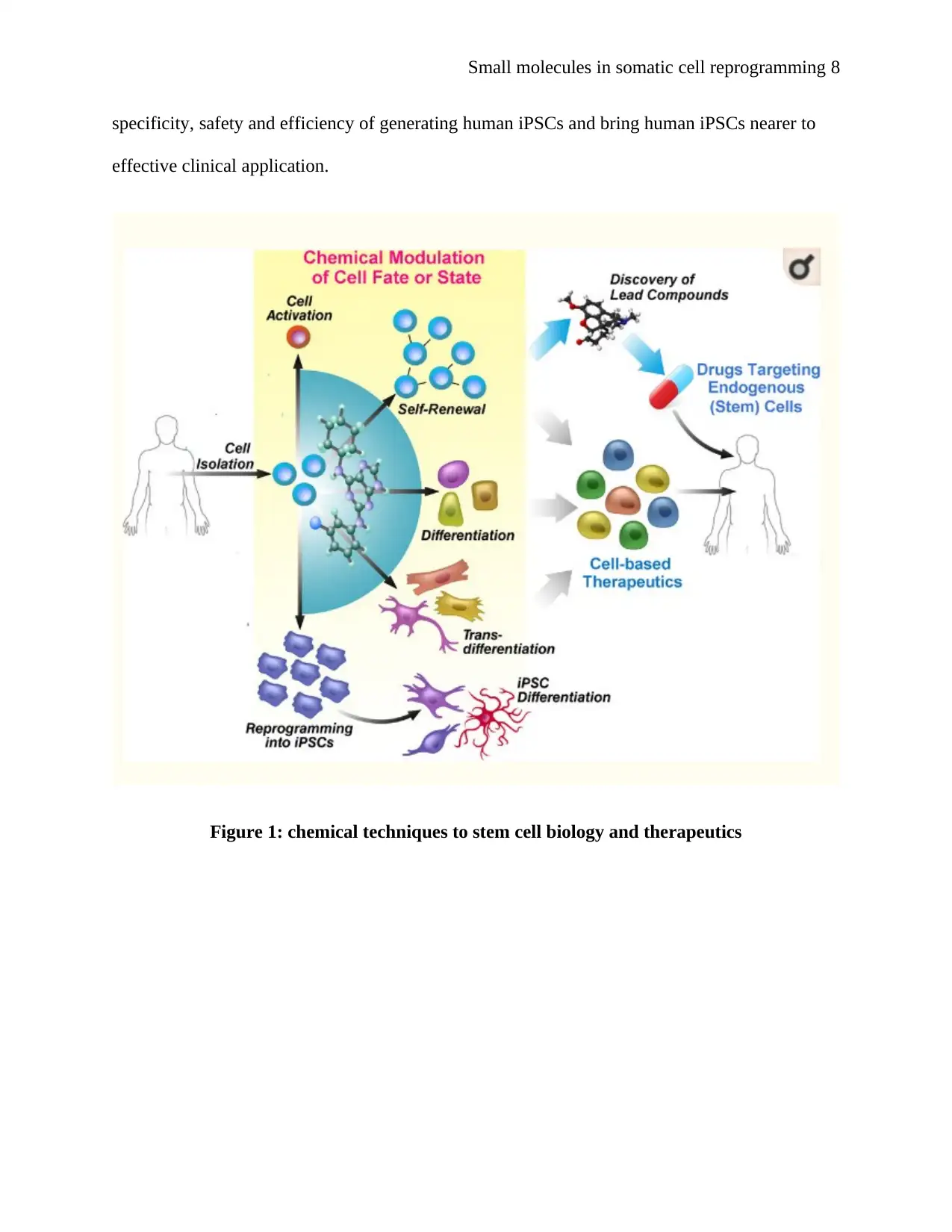
Figure 1: chemical techniques to stem cell biology and therapeutics
specificity, safety and efficiency of generating human iPSCs and bring human iPSCs nearer to
effective clinical application.
Figure 1: chemical techniques to stem cell biology and therapeutics
Small molecules in somatic cell reprogramming 9
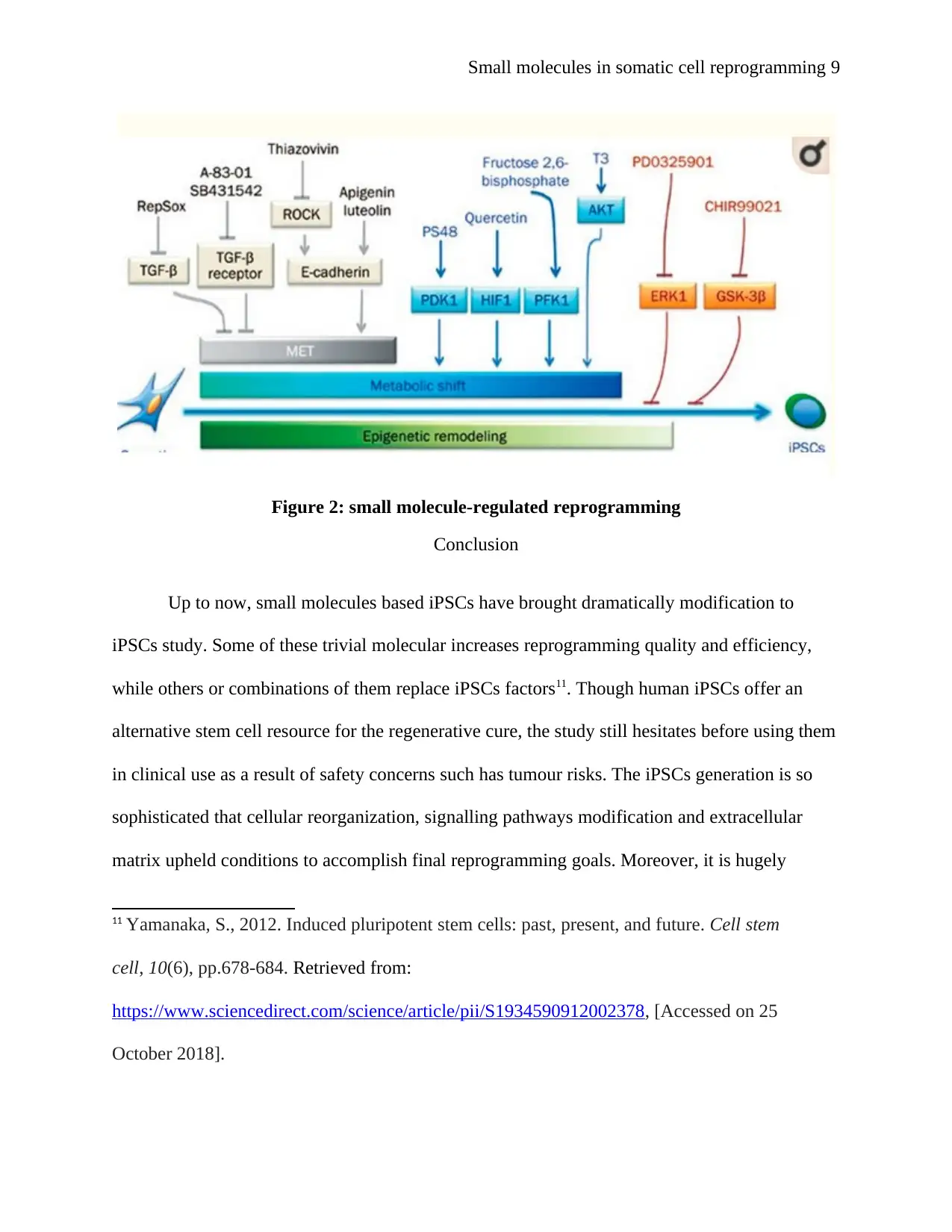
Figure 2: small molecule-regulated reprogramming
Conclusion
Up to now, small molecules based iPSCs have brought dramatically modification to
iPSCs study. Some of these trivial molecular increases reprogramming quality and efficiency,
while others or combinations of them replace iPSCs factors11. Though human iPSCs offer an
alternative stem cell resource for the regenerative cure, the study still hesitates before using them
in clinical use as a result of safety concerns such has tumour risks. The iPSCs generation is so
sophisticated that cellular reorganization, signalling pathways modification and extracellular
matrix upheld conditions to accomplish final reprogramming goals. Moreover, it is hugely
11 Yamanaka, S., 2012. Induced pluripotent stem cells: past, present, and future. Cell stem
cell, 10(6), pp.678-684. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S1934590912002378, [Accessed on 25
October 2018].
Figure 2: small molecule-regulated reprogramming
Conclusion
Up to now, small molecules based iPSCs have brought dramatically modification to
iPSCs study. Some of these trivial molecular increases reprogramming quality and efficiency,
while others or combinations of them replace iPSCs factors11. Though human iPSCs offer an
alternative stem cell resource for the regenerative cure, the study still hesitates before using them
in clinical use as a result of safety concerns such has tumour risks. The iPSCs generation is so
sophisticated that cellular reorganization, signalling pathways modification and extracellular
matrix upheld conditions to accomplish final reprogramming goals. Moreover, it is hugely
11 Yamanaka, S., 2012. Induced pluripotent stem cells: past, present, and future. Cell stem
cell, 10(6), pp.678-684. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S1934590912002378, [Accessed on 25
October 2018].
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide

Small molecules in somatic cell reprogramming 10
demanding to get highly populace of the target cells. The small molecules based technique might
offer a solution for pure target adult stem cell without tumour challenges.
The trivial molecules in the iPSCs generation might also provide important info for those
investigations in the adult stem cell. Direct reprogramming of one sort of adult stem cell to other
was proposed as an alternative for clinical application. Some studies have advanced on small
molecules that direct human transdifferentiation of tissue-specific progenitor cells.
Hematopoietic and bone marrow stem cells are prepared to be used in the medical use, and
therefore the only obstacle is their restricted propagation. Small molecules development to
function with the adult stem cell self-renewal or direct human somatic cells into progenitor cells,
will open an avenue for the hospital use of these types of progenitor and stem cells. Continuous
development of small molecular techniques and advancement in the comprehending of
reprogramming technique will facilitate the possibility of generation safety and higher quality
reprogramed cells, and also offer useful data for the adult stem cell-based use.
demanding to get highly populace of the target cells. The small molecules based technique might
offer a solution for pure target adult stem cell without tumour challenges.
The trivial molecules in the iPSCs generation might also provide important info for those
investigations in the adult stem cell. Direct reprogramming of one sort of adult stem cell to other
was proposed as an alternative for clinical application. Some studies have advanced on small
molecules that direct human transdifferentiation of tissue-specific progenitor cells.
Hematopoietic and bone marrow stem cells are prepared to be used in the medical use, and
therefore the only obstacle is their restricted propagation. Small molecules development to
function with the adult stem cell self-renewal or direct human somatic cells into progenitor cells,
will open an avenue for the hospital use of these types of progenitor and stem cells. Continuous
development of small molecular techniques and advancement in the comprehending of
reprogramming technique will facilitate the possibility of generation safety and higher quality
reprogramed cells, and also offer useful data for the adult stem cell-based use.
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

Small molecules in somatic cell reprogramming 11
References
Banito, A. and Gil, J., 2010. Induced pluripotent stem cells and senescence: learning the biology
to improve the technology. EMBO reports, 11(5), pp.353-359. Retrieved from:
http://embor.embopress.org/content/11/5/353.abstract, [Accessed on 25 October 2018].
Ichida, J.K., Blanchard, J., Lam, K., Son, E.Y., Chung, J.E., Egli, D., Loh, K.M., Carter, A.C., Di
Giorgio, F.P., Koszka, K. and Huangfu, D., 2009. A small-molecule inhibitor of Tgf-β signaling
replaces Sox2 in reprogramming by inducing Nanog. Cell stem cell, 5(5), pp.491-503. Retrieved
from: https://www.sciencedirect.com/science/article/pii/S1934590909005086, [Accessed on 25
October 2018].
Jaenisch, R., 2012. Nuclear cloning and direct reprogramming: the long and the short path to
Stockholm. Cell Stem Cell, 11(6), pp.744-747. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S193459091200639X, [Accessed on 25
October 2018].
Kamao, H., Mandai, M., Okamoto, S., Sakai, N., Suga, A., Sugita, S., Kiryu, J. and Takahashi,
M., 2014. Characterization of human induced pluripotent stem cell-derived retinal pigment
epithelium cell sheets aiming for clinical application. Stem cell reports, 2(2), pp.205-218.
Li, R., Liang, J., Ni, S., Zhou, T., Qing, X., Li, H., He, W., Chen, J., Li, F., Zhuang, Q. and Qin,
B., 2010. A mesenchymal-to-epithelial transition initiates and is required for the nuclear
reprogramming of mouse fibroblasts. Cell stem cell, 7(1), pp.51-63. Retrieved from:
https://doi.org/10.1016/j.stem.2010.04.014, [Accessed on 25 October 2018].
References
Banito, A. and Gil, J., 2010. Induced pluripotent stem cells and senescence: learning the biology
to improve the technology. EMBO reports, 11(5), pp.353-359. Retrieved from:
http://embor.embopress.org/content/11/5/353.abstract, [Accessed on 25 October 2018].
Ichida, J.K., Blanchard, J., Lam, K., Son, E.Y., Chung, J.E., Egli, D., Loh, K.M., Carter, A.C., Di
Giorgio, F.P., Koszka, K. and Huangfu, D., 2009. A small-molecule inhibitor of Tgf-β signaling
replaces Sox2 in reprogramming by inducing Nanog. Cell stem cell, 5(5), pp.491-503. Retrieved
from: https://www.sciencedirect.com/science/article/pii/S1934590909005086, [Accessed on 25
October 2018].
Jaenisch, R., 2012. Nuclear cloning and direct reprogramming: the long and the short path to
Stockholm. Cell Stem Cell, 11(6), pp.744-747. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S193459091200639X, [Accessed on 25
October 2018].
Kamao, H., Mandai, M., Okamoto, S., Sakai, N., Suga, A., Sugita, S., Kiryu, J. and Takahashi,
M., 2014. Characterization of human induced pluripotent stem cell-derived retinal pigment
epithelium cell sheets aiming for clinical application. Stem cell reports, 2(2), pp.205-218.
Li, R., Liang, J., Ni, S., Zhou, T., Qing, X., Li, H., He, W., Chen, J., Li, F., Zhuang, Q. and Qin,
B., 2010. A mesenchymal-to-epithelial transition initiates and is required for the nuclear
reprogramming of mouse fibroblasts. Cell stem cell, 7(1), pp.51-63. Retrieved from:
https://doi.org/10.1016/j.stem.2010.04.014, [Accessed on 25 October 2018].

Small molecules in somatic cell reprogramming 12
Maherali, N. and Hochedlinger, K., 2009. Tgfβ signal inhibition cooperates in the induction of
iPSCs and replaces Sox2 and cMyc. Current Biology, 19(20), pp.1718-1723. Retrieved from:
https://doi.org/10.1016/j.cub.2009.08.025, [Accessed on 25 October 2018].
Marks, H., Kalkan, T., Menafra, R., Denissov, S., Jones, K., Hofemeister, H., Nichols, J., Kranz,
A., Stewart, A.F., Smith, A. and Stunnenberg, H.G., 2012. The transcriptional and epigenomic
foundations of ground state pluripotency. Cell, 149(3), pp.590-604. Retrieved from:
https://doi.org/10.1016/j.cell.2012.03.026, [Accessed on 25 October 2018].
Nie, B., Wang, H., Laurent, T. and Ding, S., 2012. Cellular reprogramming: a small molecule
perspective. Current opinion in cell biology, 24(6), pp.784-792. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S0955067412001354, [Accessed on 25
October 2018].
Yamanaka, S., 2012. Induced pluripotent stem cells: past, present, and future. Cell stem
cell, 10(6), pp.678-684. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S1934590912002378, [Accessed on 25
October 2018].
Ying, Q.L., Wray, J., Nichols, J., Batlle-Morera, L., Doble, B., Woodgett, J., Cohen, P. and
Smith, A., 2008. The ground state of embryonic stem cell self-renewal. Nature, 453(7194),
p.519. Retrieved from: https://www.nature.com/articles/nature06968, [Accessed on 25 October
2018].
Yoshida, Y., Takahashi, K., Okita, K., Ichisaka, T. and Yamanaka, S., 2009. Hypoxia enhances
the generation of induced pluripotent stem cells. Cell stem cell, 5(3), pp.237-241. Retrieved
Maherali, N. and Hochedlinger, K., 2009. Tgfβ signal inhibition cooperates in the induction of
iPSCs and replaces Sox2 and cMyc. Current Biology, 19(20), pp.1718-1723. Retrieved from:
https://doi.org/10.1016/j.cub.2009.08.025, [Accessed on 25 October 2018].
Marks, H., Kalkan, T., Menafra, R., Denissov, S., Jones, K., Hofemeister, H., Nichols, J., Kranz,
A., Stewart, A.F., Smith, A. and Stunnenberg, H.G., 2012. The transcriptional and epigenomic
foundations of ground state pluripotency. Cell, 149(3), pp.590-604. Retrieved from:
https://doi.org/10.1016/j.cell.2012.03.026, [Accessed on 25 October 2018].
Nie, B., Wang, H., Laurent, T. and Ding, S., 2012. Cellular reprogramming: a small molecule
perspective. Current opinion in cell biology, 24(6), pp.784-792. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S0955067412001354, [Accessed on 25
October 2018].
Yamanaka, S., 2012. Induced pluripotent stem cells: past, present, and future. Cell stem
cell, 10(6), pp.678-684. Retrieved from:
https://www.sciencedirect.com/science/article/pii/S1934590912002378, [Accessed on 25
October 2018].
Ying, Q.L., Wray, J., Nichols, J., Batlle-Morera, L., Doble, B., Woodgett, J., Cohen, P. and
Smith, A., 2008. The ground state of embryonic stem cell self-renewal. Nature, 453(7194),
p.519. Retrieved from: https://www.nature.com/articles/nature06968, [Accessed on 25 October
2018].
Yoshida, Y., Takahashi, K., Okita, K., Ichisaka, T. and Yamanaka, S., 2009. Hypoxia enhances
the generation of induced pluripotent stem cells. Cell stem cell, 5(3), pp.237-241. Retrieved
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide
1 out of 13
Your All-in-One AI-Powered Toolkit for Academic Success.
+13062052269
info@desklib.com
Available 24*7 on WhatsApp / Email
![[object Object]](/_next/static/media/star-bottom.7253800d.svg)
Unlock your academic potential
Copyright © 2020–2026 A2Z Services. All Rights Reserved. Developed and managed by ZUCOL.