Water Treatment Report: Evaluation of Water Treatment Methods
VerifiedAdded on 2022/10/16
|10
|1528
|104
Report
AI Summary
This report investigates water treatment processes, focusing on water quality assessment, disinfection methods, and the impact of ferric chloride. The study evaluates the effectiveness of ferric chloride in water treatment, examining turbidity, color, DOC, and UV254 levels. It analyzes chlorine decay characteristics and THM levels, comparing different concentrations of ferric chloride. The discussion highlights the importance of disinfection in generating drinking water, the formation of disinfection byproducts, and the recommendation of limiting ferric chloride concentration to 25 mg/L to maintain acceptable THM levels. Furthermore, the report explores conventional wastewater treatment, including organic carbon removal, DO and carbon consumption, and activated sludge treatment. It explains the activated sludge process and its components, such as aeration tanks and biological floc, and the role of bacteria and algae in wastewater treatment. The report includes tables and figures to illustrate the findings and references relevant literature on water treatment and wastewater management.

Running head: WATER TREATMENT REPORT
WATER TREATMENT REPORT
Name of the Student
Name of the University
Author’s Note
WATER TREATMENT REPORT
Name of the Student
Name of the University
Author’s Note
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

1WATER TREATMENT REPORT
Table of Contents
Water treatment report...............................................................................................................2
Table 1: Water quality assessment........................................................................................2
Table 2: Optimum dose of ferric chloride..............................................................................2
Table 3: Chlorine decay characteristics for raw water...........................................................2
Table 4: THM levels in treated, raw and MQ water..............................................................2
Discussion..................................................................................................................................3
Typical conventional wastewater treatment for organic carbon removal..................................4
Table 5: DO and Carbon Consumption..................................................................................4
References..................................................................................................................................8
Table of Contents
Water treatment report...............................................................................................................2
Table 1: Water quality assessment........................................................................................2
Table 2: Optimum dose of ferric chloride..............................................................................2
Table 3: Chlorine decay characteristics for raw water...........................................................2
Table 4: THM levels in treated, raw and MQ water..............................................................2
Discussion..................................................................................................................................3
Typical conventional wastewater treatment for organic carbon removal..................................4
Table 5: DO and Carbon Consumption..................................................................................4
References..................................................................................................................................8
2WATER TREATMENT REPORT
Water treatment report
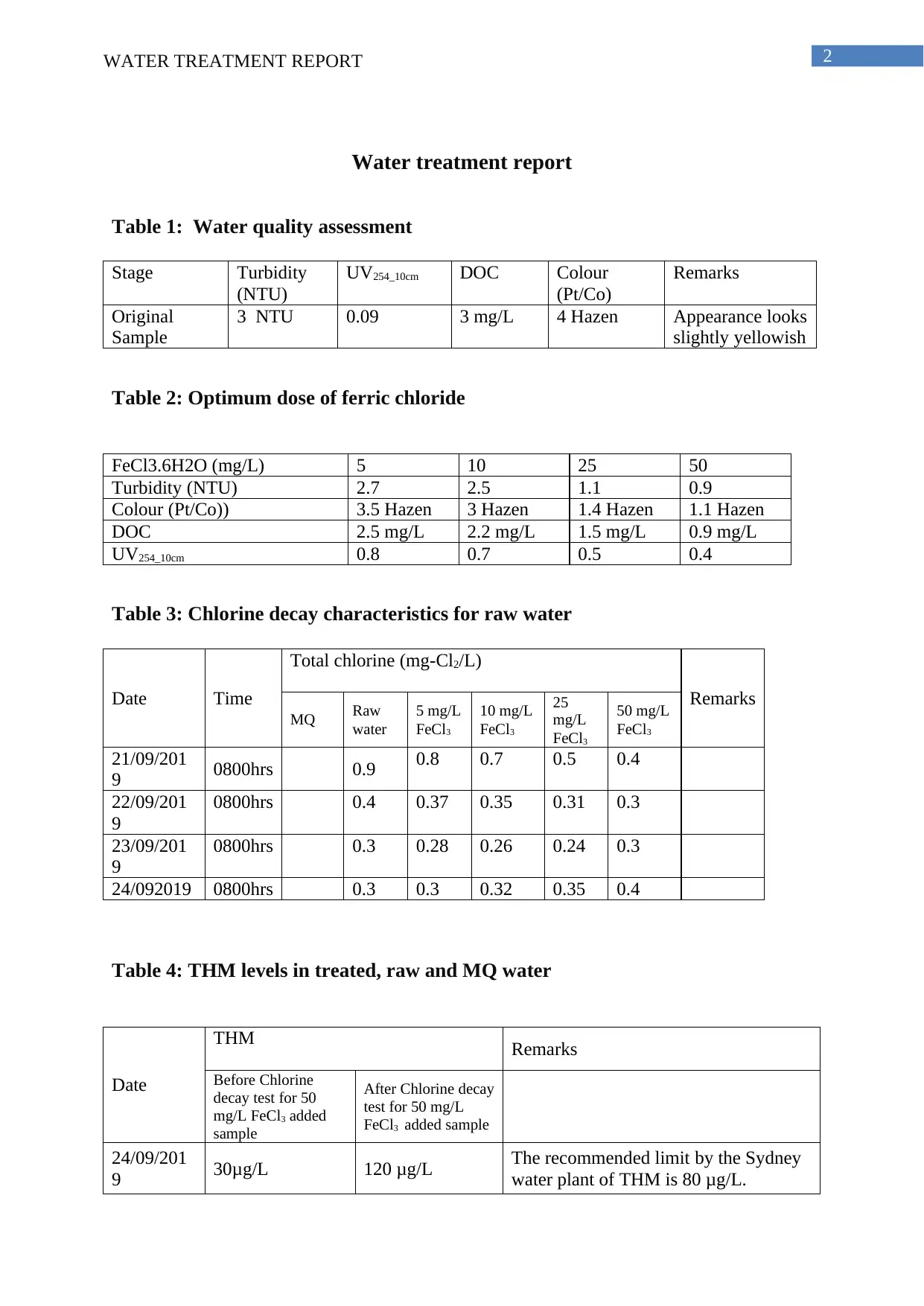
Table 1: Water quality assessment
Stage Turbidity
(NTU)
UV254_10cm DOC Colour
(Pt/Co)
Remarks
Original
Sample
3 NTU 0.09 3 mg/L 4 Hazen Appearance looks
slightly yellowish
Table 2: Optimum dose of ferric chloride
FeCl3.6H2O (mg/L) 5 10 25 50
Turbidity (NTU) 2.7 2.5 1.1 0.9
Colour (Pt/Co)) 3.5 Hazen 3 Hazen 1.4 Hazen 1.1 Hazen
DOC 2.5 mg/L 2.2 mg/L 1.5 mg/L 0.9 mg/L
UV254_10cm 0.8 0.7 0.5 0.4
Table 3: Chlorine decay characteristics for raw water
Date Time
Total chlorine (mg-Cl2/L)
Remarks
MQ Raw
water
5 mg/L
FeCl3
10 mg/L
FeCl3
25
mg/L
FeCl3
50 mg/L
FeCl3
21/09/201
9 0800hrs 0.9 0.8 0.7 0.5 0.4
22/09/201
9
0800hrs 0.4 0.37 0.35 0.31 0.3
23/09/201
9
0800hrs 0.3 0.28 0.26 0.24 0.3
24/092019 0800hrs 0.3 0.3 0.32 0.35 0.4
Table 4: THM levels in treated, raw and MQ water
Date
THM Remarks
Before Chlorine
decay test for 50
mg/L FeCl3 added
sample
After Chlorine decay
test for 50 mg/L
FeCl3 added sample
24/09/201
9 30μg/L 120 μg/L The recommended limit by the Sydney
water plant of THM is 80 μg/L.
Water treatment report
Table 1: Water quality assessment
Stage Turbidity
(NTU)
UV254_10cm DOC Colour
(Pt/Co)
Remarks
Original
Sample
3 NTU 0.09 3 mg/L 4 Hazen Appearance looks
slightly yellowish
Table 2: Optimum dose of ferric chloride
FeCl3.6H2O (mg/L) 5 10 25 50
Turbidity (NTU) 2.7 2.5 1.1 0.9
Colour (Pt/Co)) 3.5 Hazen 3 Hazen 1.4 Hazen 1.1 Hazen
DOC 2.5 mg/L 2.2 mg/L 1.5 mg/L 0.9 mg/L
UV254_10cm 0.8 0.7 0.5 0.4
Table 3: Chlorine decay characteristics for raw water
Date Time
Total chlorine (mg-Cl2/L)
Remarks
MQ Raw
water
5 mg/L
FeCl3
10 mg/L
FeCl3
25
mg/L
FeCl3
50 mg/L
FeCl3
21/09/201
9 0800hrs 0.9 0.8 0.7 0.5 0.4
22/09/201
9
0800hrs 0.4 0.37 0.35 0.31 0.3
23/09/201
9
0800hrs 0.3 0.28 0.26 0.24 0.3
24/092019 0800hrs 0.3 0.3 0.32 0.35 0.4
Table 4: THM levels in treated, raw and MQ water
Date
THM Remarks
Before Chlorine
decay test for 50
mg/L FeCl3 added
sample
After Chlorine decay
test for 50 mg/L
FeCl3 added sample
24/09/201
9 30μg/L 120 μg/L The recommended limit by the Sydney
water plant of THM is 80 μg/L.
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide

3WATER TREATMENT REPORT
Therefore, 50 mg/L FeCl3 must be
reconsidered.
Discussion
A critical treatment procedure for generating drinking water from raw natural water is
disinfection. The disinfection procedure is vital to reduce diseases and ferric chloride is
generally used as chlorine is known to disinfect water for over a century (Cadee et al., 2015).
The chlorine released from ferric chloride inactivates the microbial pathogens, however, it
reacts with other natural components of water to form by products which is not desired (NSW
Gov., 2019).
Figure 1: Procedures in waste water treatment plant summarized (Sydney water 2019)
Widespread research on improving drinking water quality standards along with
reducing the formation of disinfection byproducts (DBPs) from the chlorination of the
Therefore, 50 mg/L FeCl3 must be
reconsidered.
Discussion
A critical treatment procedure for generating drinking water from raw natural water is
disinfection. The disinfection procedure is vital to reduce diseases and ferric chloride is
generally used as chlorine is known to disinfect water for over a century (Cadee et al., 2015).
The chlorine released from ferric chloride inactivates the microbial pathogens, however, it
reacts with other natural components of water to form by products which is not desired (NSW
Gov., 2019).
Figure 1: Procedures in waste water treatment plant summarized (Sydney water 2019)
Widespread research on improving drinking water quality standards along with
reducing the formation of disinfection byproducts (DBPs) from the chlorination of the
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

4WATER TREATMENT REPORT
drinking water are been conducted. Increased intake of DBPs containing drinking water is
related to the increased risk of cancer and this malignancy is generally linked to
trihalomethanes (THMs) which is a class of DBPs byproducts (Anderson, 2013). To decrease
intake of DBP along with maintaining effectiveness of the disinfection process, the Sydney
water treatment plant recommends regulation of the THMs along with the haloacetic acids
which are all by products of FeCl3 treatment of raw natural water (Sydney water 2019).
As noted from table 3 and table 4, water disinfectant treatment with 50 mg/L FeCl3 is
not recommended as it increases the THM level beyond permissible limit (Sydney water
2019). The treatment must be restricted to 25 mg/L FeCl3 as it allows acceptable level of
THM along with proper UV254 value therefore ensuring proper disinfection of the water.
Typical conventional wastewater treatment for organic carbon removal
The carbon consumption during 1mg/L DO depletion can be calculated using as follows;
32 × 6mg/L oxygen is required to remove 12 × 6mg/L carbon
Therefore, 192mg/L oxygen is required to consume 72mg/L Carbon
250 mg/L oxygen is required to consume 94 mg/L Carbon
275 mg/L oxygen is required to consume 103 mg/L Carbon
300 mg/L oxygen is required to consume 112 mg/L Carbon and so on
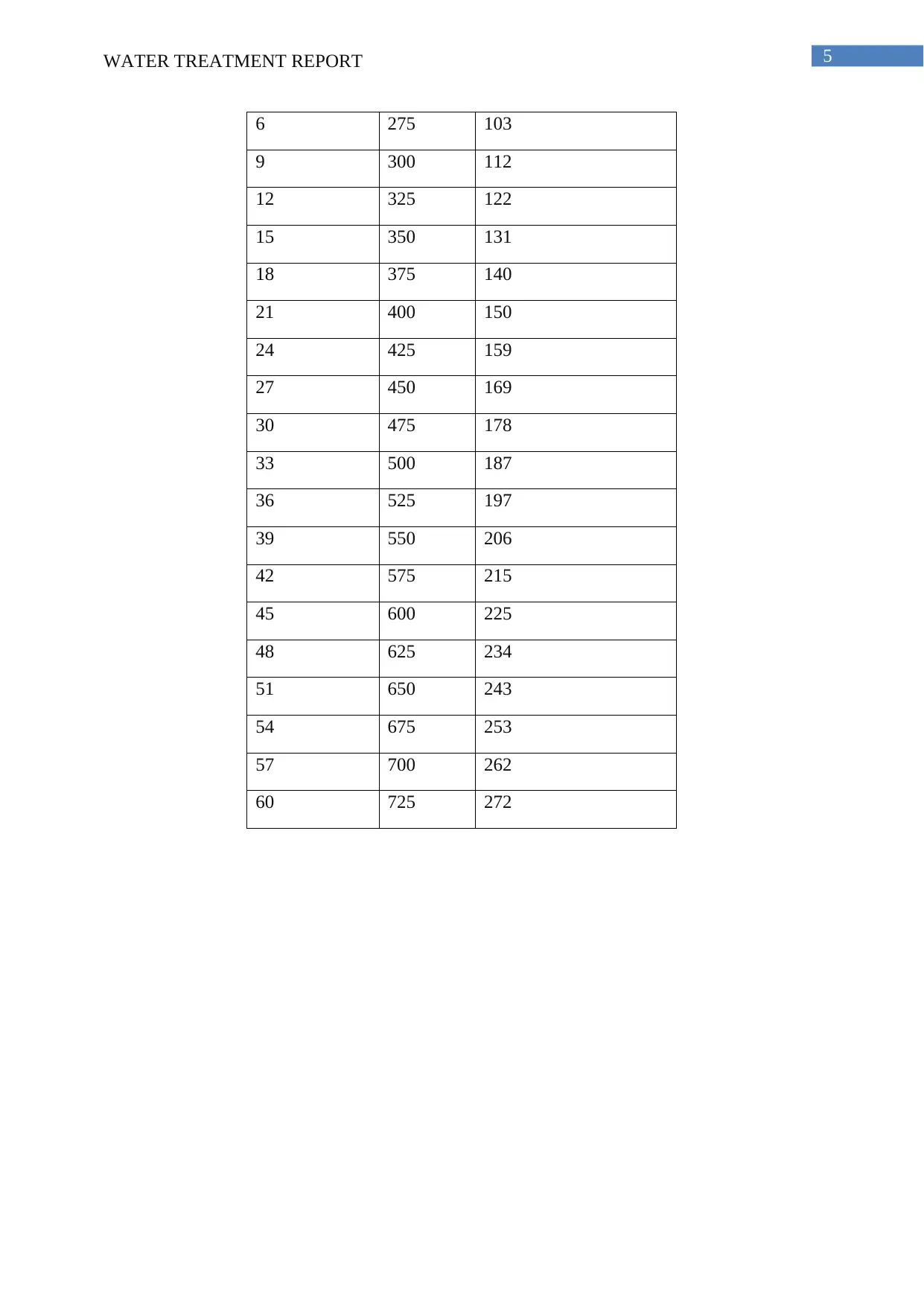
Table 5: DO and Carbon Consumption
Time (minutes) DO(mg/
L) Carbon Removal (mg/L)
0 192 72
3 250 94
drinking water are been conducted. Increased intake of DBPs containing drinking water is
related to the increased risk of cancer and this malignancy is generally linked to
trihalomethanes (THMs) which is a class of DBPs byproducts (Anderson, 2013). To decrease
intake of DBP along with maintaining effectiveness of the disinfection process, the Sydney
water treatment plant recommends regulation of the THMs along with the haloacetic acids
which are all by products of FeCl3 treatment of raw natural water (Sydney water 2019).
As noted from table 3 and table 4, water disinfectant treatment with 50 mg/L FeCl3 is
not recommended as it increases the THM level beyond permissible limit (Sydney water
2019). The treatment must be restricted to 25 mg/L FeCl3 as it allows acceptable level of
THM along with proper UV254 value therefore ensuring proper disinfection of the water.
Typical conventional wastewater treatment for organic carbon removal
The carbon consumption during 1mg/L DO depletion can be calculated using as follows;
32 × 6mg/L oxygen is required to remove 12 × 6mg/L carbon
Therefore, 192mg/L oxygen is required to consume 72mg/L Carbon
250 mg/L oxygen is required to consume 94 mg/L Carbon
275 mg/L oxygen is required to consume 103 mg/L Carbon
300 mg/L oxygen is required to consume 112 mg/L Carbon and so on
Table 5: DO and Carbon Consumption
Time (minutes) DO(mg/
L) Carbon Removal (mg/L)
0 192 72
3 250 94
5WATER TREATMENT REPORT
6 275 103
9 300 112
12 325 122
15 350 131
18 375 140
21 400 150
24 425 159
27 450 169
30 475 178
33 500 187
36 525 197
39 550 206
42 575 215
45 600 225
48 625 234
51 650 243
54 675 253
57 700 262
60 725 272
6 275 103
9 300 112
12 325 122
15 350 131
18 375 140
21 400 150
24 425 159
27 450 169
30 475 178
33 500 187
36 525 197
39 550 206
42 575 215
45 600 225
48 625 234
51 650 243
54 675 253
57 700 262
60 725 272
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide
6WATER TREATMENT REPORT
0 3 6 9 12 15 18 21 24 27 30 33 36 39 42 45 48 51 54 57 60
192
250 275 300 325 350 375 400 425 450 475 500 525 550 575 600 625 650 675 700 725
72 94 103 112 122 131 140 150 159 169 178 187 197 206 215 225 234 243 253 262 272
DO(mg/L) Carbon Removal (mg/L)
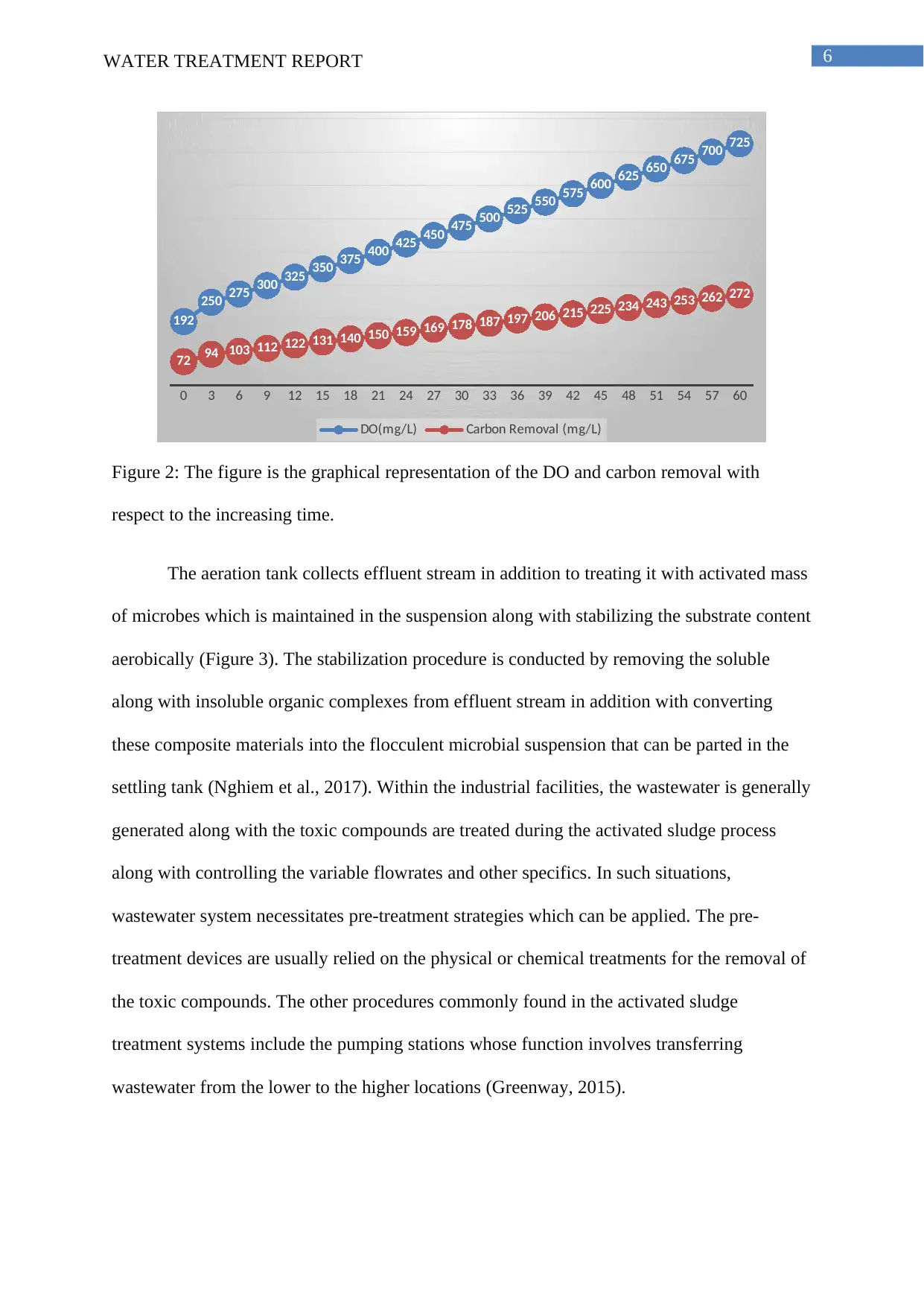
Figure 2: The figure is the graphical representation of the DO and carbon removal with
respect to the increasing time.
The aeration tank collects effluent stream in addition to treating it with activated mass
of microbes which is maintained in the suspension along with stabilizing the substrate content
aerobically (Figure 3). The stabilization procedure is conducted by removing the soluble
along with insoluble organic complexes from effluent stream in addition with converting
these composite materials into the flocculent microbial suspension that can be parted in the
settling tank (Nghiem et al., 2017). Within the industrial facilities, the wastewater is generally
generated along with the toxic compounds are treated during the activated sludge process
along with controlling the variable flowrates and other specifics. In such situations,
wastewater system necessitates pre-treatment strategies which can be applied. The pre-
treatment devices are usually relied on the physical or chemical treatments for the removal of
the toxic compounds. The other procedures commonly found in the activated sludge
treatment systems include the pumping stations whose function involves transferring
wastewater from the lower to the higher locations (Greenway, 2015).
0 3 6 9 12 15 18 21 24 27 30 33 36 39 42 45 48 51 54 57 60
192
250 275 300 325 350 375 400 425 450 475 500 525 550 575 600 625 650 675 700 725
72 94 103 112 122 131 140 150 159 169 178 187 197 206 215 225 234 243 253 262 272
DO(mg/L) Carbon Removal (mg/L)
Figure 2: The figure is the graphical representation of the DO and carbon removal with
respect to the increasing time.
The aeration tank collects effluent stream in addition to treating it with activated mass
of microbes which is maintained in the suspension along with stabilizing the substrate content
aerobically (Figure 3). The stabilization procedure is conducted by removing the soluble
along with insoluble organic complexes from effluent stream in addition with converting
these composite materials into the flocculent microbial suspension that can be parted in the
settling tank (Nghiem et al., 2017). Within the industrial facilities, the wastewater is generally
generated along with the toxic compounds are treated during the activated sludge process
along with controlling the variable flowrates and other specifics. In such situations,
wastewater system necessitates pre-treatment strategies which can be applied. The pre-
treatment devices are usually relied on the physical or chemical treatments for the removal of
the toxic compounds. The other procedures commonly found in the activated sludge
treatment systems include the pumping stations whose function involves transferring
wastewater from the lower to the higher locations (Greenway, 2015).
Paraphrase This Document
Need a fresh take? Get an instant paraphrase of this document with our AI Paraphraser

7WATER TREATMENT REPORT
Figure 3: Activated sludge treatment procedure (Gouveia & Pinto, 2014)
The treatment of activated sludge is kind of a wastewater treatment procedure for the
treatment of wastewater with the aid of aeration in addition to the biological floc integrated
into the water which is composed of selected bacteria and algae (Figure 3). The organic
matter suspended or dissolved in water is metabolized by the heterotrophic bacteria added for
processing of water which also involves uptake of the available oxygen in water during the
conventional biological aerobic wastewater management processes (Inglis et al., 2015). This
depleted oxygen is replaced with aeration process as regular water treatment plants lack
sufficient photosynthetic plants which would replace the depleted oxygen (Greenway &
Simpson, 2016). High temperature along with ample sunlight generate suitable environment
which encourages algal growth which could utilize the extra carbon dioxide which is released
by the bacteria during the breakdown of the organic components present in wastewater along
with nitrogen and phosphorus processing which are present in wastewater (Greenway, 2015).
The result illustrated in Figure 2 highlights the proper functioning of the concerned activated
sludge of the water treatment plant as consistent increase of dissolved oxygen (DO) and
carbon removal can be noted with increasing time.
Figure 3: Activated sludge treatment procedure (Gouveia & Pinto, 2014)
The treatment of activated sludge is kind of a wastewater treatment procedure for the
treatment of wastewater with the aid of aeration in addition to the biological floc integrated
into the water which is composed of selected bacteria and algae (Figure 3). The organic
matter suspended or dissolved in water is metabolized by the heterotrophic bacteria added for
processing of water which also involves uptake of the available oxygen in water during the
conventional biological aerobic wastewater management processes (Inglis et al., 2015). This
depleted oxygen is replaced with aeration process as regular water treatment plants lack
sufficient photosynthetic plants which would replace the depleted oxygen (Greenway &
Simpson, 2016). High temperature along with ample sunlight generate suitable environment
which encourages algal growth which could utilize the extra carbon dioxide which is released
by the bacteria during the breakdown of the organic components present in wastewater along
with nitrogen and phosphorus processing which are present in wastewater (Greenway, 2015).
The result illustrated in Figure 2 highlights the proper functioning of the concerned activated
sludge of the water treatment plant as consistent increase of dissolved oxygen (DO) and
carbon removal can be noted with increasing time.

8WATER TREATMENT REPORT
References
Anderson, J. (2013). The environmental benefits of water recycling and reuse. Water Science
and Technology: Water Supply, 3(4), 1-10.
Cadee, K., O’Leary, B., Smith, P., Slunjski, M., & Bourke, M. (2015, June). World’s first
magnetic ion exchange (MIEX®) water treatment plant to be installed in Western
Australia. In Orıca Watercare, Australian Water Works Association Conference,
Denver, USA, Proceedings (pp. 11-15).
Greenway, M. (2015). The role of constructed wetlands in secondary effluent treatment and
water reuse in subtropical and arid Australia. Ecological Engineering, 25(5), 501-509.
Greenway, M., & Simpson, J. S. (2016). Artificial wetlands for wastewater treatment, water
reuse and wildlife in Queensland, Australia. Water Science and Technology, 33(10-
11), 221-229.
Gouveia, R., & Pinto, J. M. (2014). Optimal policies for activated sludge treatment systems
with multi effluent stream generation. Brazilian Journal of Chemical
Engineering, 17(4-7), 979-990.
Inglis, T. J., Garrow, S. C., Henderson, M., Clair, A., Sampson, J., O'Reilly, L., & Cameron,
B. (2015). Burkholderia pseudomallei traced to water treatment plant in
Australia. Emerging infectious diseases, 6(1), 56.
Nghiem, L. D., Ren, T., Aziz, N., Porter, I., & Regmi, G. (2017). Treatment of coal seam gas
produced water for beneficial use in Australia: a review of best
practices. Desalination and Water Treatment, 32(1-3), 316-323.
References
Anderson, J. (2013). The environmental benefits of water recycling and reuse. Water Science
and Technology: Water Supply, 3(4), 1-10.
Cadee, K., O’Leary, B., Smith, P., Slunjski, M., & Bourke, M. (2015, June). World’s first
magnetic ion exchange (MIEX®) water treatment plant to be installed in Western
Australia. In Orıca Watercare, Australian Water Works Association Conference,
Denver, USA, Proceedings (pp. 11-15).
Greenway, M. (2015). The role of constructed wetlands in secondary effluent treatment and
water reuse in subtropical and arid Australia. Ecological Engineering, 25(5), 501-509.
Greenway, M., & Simpson, J. S. (2016). Artificial wetlands for wastewater treatment, water
reuse and wildlife in Queensland, Australia. Water Science and Technology, 33(10-
11), 221-229.
Gouveia, R., & Pinto, J. M. (2014). Optimal policies for activated sludge treatment systems
with multi effluent stream generation. Brazilian Journal of Chemical
Engineering, 17(4-7), 979-990.
Inglis, T. J., Garrow, S. C., Henderson, M., Clair, A., Sampson, J., O'Reilly, L., & Cameron,
B. (2015). Burkholderia pseudomallei traced to water treatment plant in
Australia. Emerging infectious diseases, 6(1), 56.
Nghiem, L. D., Ren, T., Aziz, N., Porter, I., & Regmi, G. (2017). Treatment of coal seam gas
produced water for beneficial use in Australia: a review of best
practices. Desalination and Water Treatment, 32(1-3), 316-323.
⊘ This is a preview!⊘
Do you want full access?
Subscribe today to unlock all pages.

Trusted by 1+ million students worldwide

9WATER TREATMENT REPORT
NSW Gov. 2019. NSW Government Australian Drinking Water Guidelines 2011 Available at
https://www.health.nsw.gov.au/environment/water/Pages/drinking-water.aspx
Sydney water 2019. Cronulla wastewater treatment. Available at
https://www.sydneywater.com.au/SW/education/Wastewater-recycling/Wastewater-
treatment/cronulla-wastewater-treatment/index.htm#
NSW Gov. 2019. NSW Government Australian Drinking Water Guidelines 2011 Available at
https://www.health.nsw.gov.au/environment/water/Pages/drinking-water.aspx
Sydney water 2019. Cronulla wastewater treatment. Available at
https://www.sydneywater.com.au/SW/education/Wastewater-recycling/Wastewater-
treatment/cronulla-wastewater-treatment/index.htm#
1 out of 10
Your All-in-One AI-Powered Toolkit for Academic Success.
+13062052269
info@desklib.com
Available 24*7 on WhatsApp / Email
![[object Object]](/_next/static/media/star-bottom.7253800d.svg)
Unlock your academic potential
Copyright © 2020–2026 A2Z Services. All Rights Reserved. Developed and managed by ZUCOL.

